Targetable immune pathway plays an early role in Alzheimer’s disease models, before plaque accumulation.
Researchers at Boston Children’s Hospital show how brain connections, or synapses, are lost early in Alzheimer’s disease and demonstrate that the process starts, and could potentially be halted, before telltale plaques accumulate in the brain. Their work, published online by Science on March 31, suggests new therapeutic targets to preserve cognitive function early in Alzheimer’s disease.
Led by Beth Stevens, PhD, and Soyon Hong, PhD, of Boston Children’s F.M. Kirby Neurobiology Center, the researchers show in multiple mouse models of Alzheimer’s that mechanisms similar to those used to “prune” excess synapses in the healthy developing brain are wrongly activated later in life. By blocking these mechanisms, they were able to reduce synapse loss in the mice.
Currently, there are five FDA-approved drugs for Alzheimer’s, but these only boost cognition temporarily and do not address the root causes of cognitive impairment in Alzheimer’s. Many newer drugs in the pipeline seek to eliminate amyloid plaque deposits or reduce inflammation in the brain, but the new research from Boston Children’s suggests that Alzheimer’s could be targeted much earlier, before these pathologic changes occur.
“Synapse loss is a strong correlate of cognitive decline,” says Stevens, assistant professor in the Department of Neurology at Boston Children’s, senior investigator on the study and a recent recipient of the MacArthur “genius” grant. “We’re trying to go back to the very beginning and see how synapse loss starts.”
The young brain informing the old
Stevens, Hong and colleagues looked at Alzheimer’s, a disease of aging, through an unusual lens: normal brain development in infancy and childhood. Through years of research, the Stevens lab has shown that normal developing brains have a process to “prune” synapses that aren’t needed as they build their circuitry. (See accompanying backgrounder.)
“Understanding a normal developmental process deeply has provided us with novel insight into how to protect synapses in Alzheimer’s and potentially a host of other diseases,” says Stevens, noting that synapse loss also occurs in frontotemporal dementia, Huntington’s disease, schizophrenia, glaucoma and other conditions.
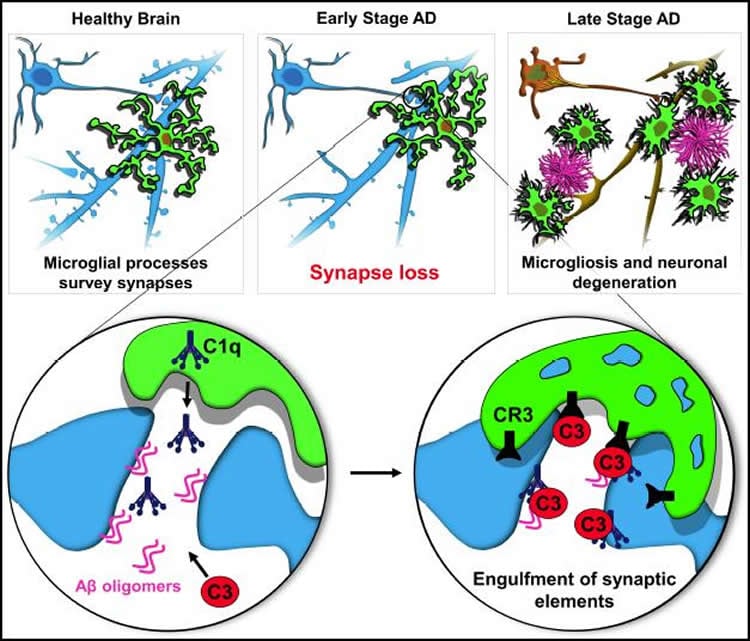
In the Alzheimer’s mouse models, the team showed that synapse loss requires the activation of a protein called C1q, which “tags” synapses for elimination. Immune cells in the brain called microglia then “eat” the synapses — similar to what occurs during normal brain development. In the mice, C1q became more abundant around vulnerable synapses before amyloid plaque deposits could be observed.
Therapeutic potential
When Stevens and colleagues blocked C1q, a downstream protein called C3, or the C3 receptor on microglia, synapse loss did not occur.
“Microglia and complement are already known to be involved in Alzheimer’s disease, but they have been largely regarded as a secondary event related to plaque-related neuroinflammation, a prominent feature in progressed stages of Alzheimer’s,” notes Hong, the Science paper’s first author. “Our study challenges this view and provides evidence that complement and microglia are involved much earlier in the disease process, when synapses are already vulnerable, and could potentially be targeted to preserve synaptic health.”
A human form of the antibody Stevens and Hong used to block C1q, known as ANX-005, is in early therapeutic development with Annexon Biosciences (San Francisco) and is being advanced into the clinic. The researchers believe it has potential to be used someday to protect against synapse loss in a variety of neurodegenerative diseases.
“One of the things this study highlights is the need to look for biomarkers for synapse loss and dysfunction,” says Hong. “As in cancer, if you treat people at a later stage of Alzheimer’s, it may already be too late.”
Complement, microglia and beta-amyloid
Together with co-author Dennis Selkoe, MD, at Brigham and Women’s Hospital, the researchers also found that that beta-amyloid protein, C1q and microglia work together to cause synapse loss in the early stages of Alzheimer’s. The oligomeric form of beta-amyloid (multiple units of beta-amyloid strung together) is already known to be toxic to synapses even before it forms plaque deposits, but the study showed that C1q is necessary for this effect. The converse was also true: microglia engulfed synapses only when oligomeric beta-amyloid was present.
Annexon co-founders Ben Barres, MD, PhD, and Arnon Rosenthal, PhD, are coauthors on the paper. Barres, Rosenthal and Stevens are minor shareholders of Annexon LLC and Stevens is a member of the Scientific Advisory Board.
Funding: The study was funded by Edward R. and Anne G. Lefler Fellowship, Coins for Alzheimer’s Research Trust, Fidelity Biosciences Research Initiative (F-Prime), JPB Foundation and the National Institutes of Health (AG000222), NINDS-NIH R01NS083845 and the NIH National Institute on Aging (1RF1AG051496A).
Source: Bethany Tripp – Boston Children’s Hospital
Image Source: The image is credited to Lasse Dissing-Olesen, Boston Children’s Hospital.
Original Research: Abstract for “Complement and microglia mediate early synapse loss in Alzheimer mouse models” by Soyon Hong, Victoria F. Beja-Glasser, Bianca M. Nfonoyim, Arnaud Frouin, Shaomin Li, Saranya Ramakrishnan, Katherine M. Merry, Qiaoqiao Shi, Arnon Rosenthal, Ben A. Barres, Cynthia A. Lemere, Dennis J. Selkoe, and Beth Stevens in Science. Published online March 31 2016 doi:10.1126/science.aad8373
Abstract
Complement and microglia mediate early synapse loss in Alzheimer mouse models
Synapse loss in Alzheimer’s disease (AD) correlates with cognitive decline. Involvement of microglia and complement in AD has been attributed to neuroinflammation, prominent late in disease. Here we show in mouse models that complement and microglia mediate synaptic loss early in AD. C1q, the initiating protein of the classical complement cascade, is increased and associated with synapses before overt plaque deposition. Inhibition of C1q, C3 or the microglial complement receptor CR3, reduces the number of phagocytic microglia as well as the extent of early synapse loss. C1q is necessary for the toxic effects of soluble β-amyloid (Aβ) oligomers on synapses and hippocampal long-term potentiation (LTP). Finally, microglia in adult brains engulf synaptic material in a CR3-dependent process when exposed to soluble Aβ oligomers. Together, these findings suggest that the complement-dependent pathway and microglia that prune excess synapses in development are inappropriately activated and mediate synapse loss in AD.
“Complement and microglia mediate early synapse loss in Alzheimer mouse models” by Soyon Hong, Victoria F. Beja-Glasser, Bianca M. Nfonoyim, Arnaud Frouin, Shaomin Li, Saranya Ramakrishnan, Katherine M. Merry, Qiaoqiao Shi, Arnon Rosenthal, Ben A. Barres, Cynthia A. Lemere, Dennis J. Selkoe, and Beth Stevens in Science. Published online March 31 2016 doi:10.1126/science.aad8373






