Fat decreases glucose levels in the mouse brain.
An international research team has now investigated their interactions in different states of consciousness and has discovered that patients with severely impaired consciousness (disorders of consciousness, e.g. vegetative state/unresponsive wakefulness syndrome and minimally conscious state) show a pathological or uncontrolled communication between the two networks. Conversely, in patients who recovered from disorders of consciousness (patients known as emerging from minimally conscious state) the habitual interactions between networks are partially preserved, despite there being no difference in the connectivity within each network.
A high-fat diet of three days in mice leads to a reduction in the amount of glucose that reaches the brain. This finding was reported by a Research Group led by Jens Brüning, Director at the Max Planck Institute for Metabolism Research in Cologne. The mouse brain restored its sugar uptake after four weeks, albeit at the cost of the rest of the body.
High-fat-content foods throw our bodies out of kilter. Obesity and diseases such as type 2 diabetes can be the result. But what does a high-fat diet actually do to our brain? Scientists from the Max Planck Institute for Metabolism Research in Cologne have looked into the brains of mice to understand how obesity and diabetes develop.
“A high-fat diet reduces the uptake of blood glucose into the brain in as little as three days. So the brain is starving, even though the mice are consuming a lot of calories daily. Responsible for this is the protein GLUT-1, which is the most important glucose transporter at the blood-brain barrier,” explained Alexander Jais, author of the study. Possible triggers for the reduction of the GLUT-1 transporter are free saturated fatty acids that have a toxic effect on the cells of the blood-brain barrier. The brain lacks glucose in significant areas: the hypothalamus, which controls metabolism, and the cerebral cortex, responsible for learning and memory.
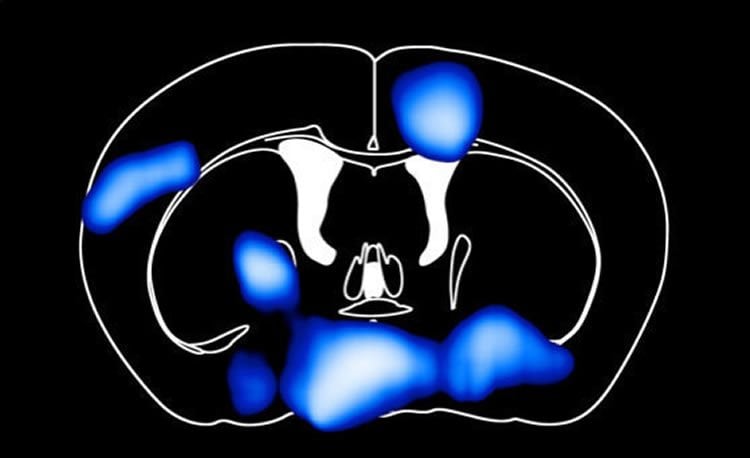
The brain takes action to compensate for its lack of energy. Macrophages, specialized cells in the immune system, produce the growth factor VEGF, which boosts the production and function of GLUT-1, and release it directly at the vascular endothelial cells of the blood-brain barrier. Normal glucose levels can then be measured in the brain after four weeks, even though the mice continue to eat a high-fat diet. If the mice have no VEGF, glucose uptake in the brain remains reduced. “As a result of this, the mice learn more slowly and have impaired memory,” said Jais.
The selfish brain
Balancing the brain’s need for sugar when the diet remains high in fat can only be done at the cost of the rest of the body. “We call it the selfish brain, because it gets its glucose by stimulating the body’s appetite for sweet foods and in the long run preventing the uptake in muscles and fat. The cells in the musculature are becoming resistant to insulin, the hormone that normally regulates glucose uptake in the cells of these organs. Ultimately, this leads to the development of diabetes,” explained Jais.
Source: Maren Berghoff – Max Planck Institute
Image Source: The image is credited to MPI f. Metabolism Research.
Original Research: Abstract for “Myeloid-Cell-Derived VEGF maintains brain glucose uptake and limits cognitive impairment in obesity” by Alexander Jais, Maite Solas, Heiko Backes, Bhagirath Chaurasia, André Kleinridders, Sebastian Theurich, Jan Mauer, Sophie M. Steculorum, Brigitte Hampel, Julia Goldau, Jens Alber, Carola Y. Förster, Sabine A. Eming, Markus Schwaninger, Napoleone Ferrara, Gerard Karsenty, and Jens C. Brüning in Cell. Published online April 28 2016 doi:10.1016/j.cell.2016.03.033
Abstract
Myeloid-Cell-Derived VEGF maintains brain glucose uptake and limits cognitive impairment in obesity
Highlights
•Acute high-fat feeding suppresses GLUT1 expression at the blood-brain barrier (BBB)
•Macrophages at the BBB increase VEGF expression upon prolonged HFD feeding
•Inducible GLUT1 deletion in brain endothelial cells leads to increased VEGF secretion
•Myeloid-cell-specific disruption of VEGF impairs cognitive function in obesity
Summary
High-fat diet (HFD) feeding induces rapid reprogramming of systemic metabolism. Here, we demonstrate that HFD feeding of mice downregulates glucose transporter (GLUT)-1 expression in blood-brain barrier (BBB) vascular endothelial cells (BECs) and reduces brain glucose uptake. Upon prolonged HFD feeding, GLUT1 expression is restored, which is paralleled by increased expression of vascular endothelial growth factor (VEGF) in macrophages at the BBB. In turn, inducible reduction of GLUT1 expression specifically in BECs reduces brain glucose uptake and increases VEGF serum concentrations in lean mice. Conversely, myeloid-cell-specific deletion of VEGF in VEGFΔmyel mice impairs BBB-GLUT1 expression, brain glucose uptake, and memory formation in obese, but not in lean mice. Moreover, obese VEGFΔmyel mice exhibit exaggerated progression of cognitive decline and neuroinflammation on an Alzheimer’s disease background. These experiments reveal that transient, HFD-elicited reduction of brain glucose uptake initiates a compensatory increase of VEGF production and assign obesity-associated macrophage activation a homeostatic role to restore cerebral glucose metabolism, preserve cognitive function, and limit neurodegeneration in obesity.
“Myeloid-Cell-Derived VEGF maintains brain glucose uptake and limits cognitive impairment in obesity” by Alexander Jais, Maite Solas, Heiko Backes, Bhagirath Chaurasia, André Kleinridders, Sebastian Theurich, Jan Mauer, Sophie M. Steculorum, Brigitte Hampel, Julia Goldau, Jens Alber, Carola Y. Förster, Sabine A. Eming, Markus Schwaninger, Napoleone Ferrara, Gerard Karsenty, and Jens C. Brüning in Cell. Published online April 28 2016 doi:10.1016/j.cell.2016.03.033







