Summary: Researchers have discovered how TH1 and TH17 immune cells obtain access to neurons in multiple sclerosis.
Source: University of Illinois at Chicago.
Drug designers working on therapeutics against multiple sclerosis should focus on blocking two distinct ways rogue immune cells attack healthy neurons, according to a new study in the journal Cell Reports.
In multiple sclerosis, immune cells degrade the insulation that protects neurons and allows them to signal to one another, but little is known about how immune cells penetrate the blood-brain barrier to get to neurons. Researchers led by Sarah Lutz, University of Illinois at Chicago College of Medicine while she was a post-doctoral fellow of Dritan Agalliu at Columbia University; and Sunil Gandhi, University of California, Irvine, have uncovered two different ways immune cells gain access to neurons and wreak their havoc.
Multiple sclerosis is a neurodegenerative inflammatory disease that affects approximately 2.5 million people worldwide. Immune cells turn against the body and cause damage to the myelin sheath, which encases neurons like the insulation on a wire. Loss of myelin interferes with the transmission of signals along the nerve fibers and impairs motor function, including walking and speech. Symptoms, which can be sporadic or progressive, range from mild to debilitating.
While researchers have known that two different kinds of immune cells, Th1 and Th17 lymphocytes, are involved in degrading myelin around neurons in multiple sclerosis, they didn’t know exactly how these cells crossed the blood-brain barrier to access neurons.
The blood-brain barrier is a bit of a misnomer. It not only protects the brain, but also the spine, and refers to the fact that blood vessels that supply the brain and spine are virtually impermeable because the cells that make up those blood vessels — called endothelial cells — are bolted tightly together by protein complexes called tight junctions. This prevents certain chemicals, harmful microbes and cells that circulate in the blood from gaining access to the brain and spine. In blood vessels that supply other organs of the body, endothelial cells are more loosely bound to one another and the connections can be adjusted to allow for the exchange of molecules and cells from the bloodstream into tissues and vice versa.
“In autoimmune diseases like multiple sclerosis, immune cells that enter the brain and spinal cord cause disease,” said Lutz, assistant professor of anatomy and cell biology in the UIC College of Medicine and the lead author of the paper. “A better understanding of how these cells cross the blood-brain barrier will aid our efforts to develop specific therapies to keep them out.”
To explore how Th1 and Th17 immune cells gain access to neurons in multiple sclerosis, Lutz and her colleagues looked at the blood-brain barrier in mice with experimental autoimmune encephalomyelitis — a mouse version of multiple sclerosis.
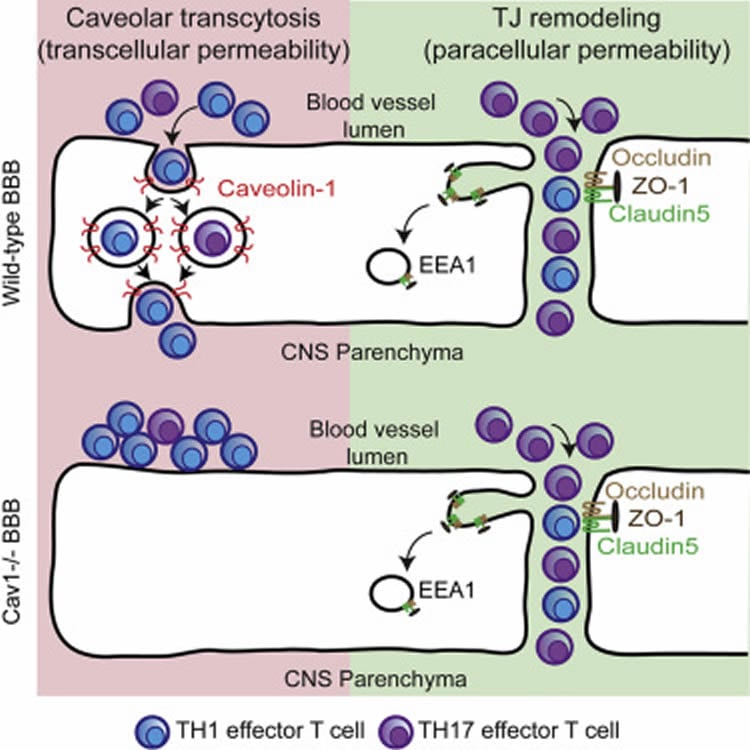
They genetically labeled blood vessel endothelial cell tight junctions with a fluorescent protein to examine if and how tight junctions are involved in autoimmune encephalomyelitis in vivo in their mice. The researchers observed that the tight junctions were significantly deteriorated in the presence of Th17 cells, and that this took place early in the onset of disease. Approximately three days later in the disease process, Lutz and colleagues found that Th1 cells were accessing and degrading myelin and neurons — but these cells did not pass through tight junctions like the Th17 cells did. Instead, the circulating Th1 cells got to neurons by going through the blood vessel endothelial cells using specialized cell membrane structures called caveolae. Caveolae are small pits or “caves” found on the surface of many cell types and help facilitate the passage of various molecules and cells into and/or through cells. In mice with autoimmune encephalomyelitis bred to lack caveolae, the researchers found almost no Th1 cells in the brain and spinal cord. They determined that caveolae on endothelial cells that make up blood vessels are required to help ferry Th1 cells through the blood-brain barrier.
“This is the first time we have ever seen, in live animals in real-time, the different means by which these two cell types gain access to myelin and nerves,” said Lutz. “Now that we know how these cells get to neurons, drugs or small molecules can be designed that interfere with or block each of these processes to help treat and possibly prevent multiple sclerosis.”
Funding: This research was supported by grants R01 HL116995, R56 MH109987 and R01 MH112849 from the National Institutes of Health; grants MNSS RG4673A1/1 and FG2035-A-1 from the National Multiple Sclerosis Society, and funds from the Leducq Foundation, and from John Castle to the department of neurology and stroke division at the Columbia University Medical Center.
Dae Hwan Kim, Carl V. L. Olson, Kyle Ellefsen, and Jennifer Bates, University of California, Irvine; and Julian Smith, Columbia University Medical Center, New York, are co-authors on the paper.
Source: Sharon Parmet – University of Illinois at Chicago
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Agalliu et al./Cell Reports.
Original Research: Full open access research for “Caveolin1 Is Required for Th1 Cell Infiltration, but Not Tight Junction Remodeling, at the Blood-Brain Barrier in Autoimmune Neuroinflammation” by Sarah E. Lutz, Julian R. Smith, Dae Hwan Kim, Carl V.L. Olson, Kyle Ellefsen, Jennifer M. Bates, Sunil P. Gandhi, and Dritan Agalliu in Cell Reports. Published online November 21 2017 doi:10.1016/j.celrep.2017.10.094
[cbtabs][cbtab title=”MLA”]University of Illinois at Chicago “How Rogue Immune Cells Cross the Blood-Brain Barrier to Cause Multiple Sclerosis.” NeuroscienceNews. NeuroscienceNews, 22 November 2017.
<https://neurosciencenews.com/ms-immune-cells-bbb-8011/>.[/cbtab][cbtab title=”APA”]University of Illinois at Chicago (2017, November 22). How Rogue Immune Cells Cross the Blood-Brain Barrier to Cause Multiple Sclerosis. NeuroscienceNews. Retrieved November 22, 2017 from https://neurosciencenews.com/ms-immune-cells-bbb-8011/[/cbtab][cbtab title=”Chicago”]University of Illinois at Chicago “How Rogue Immune Cells Cross the Blood-Brain Barrier to Cause Multiple Sclerosis.” https://neurosciencenews.com/ms-immune-cells-bbb-8011/ (accessed November 22, 2017).[/cbtab][/cbtabs]
Abstract
Caveolin1 Is Required for Th1 Cell Infiltration, but Not Tight Junction Remodeling, at the Blood-Brain Barrier in Autoimmune Neuroinflammation
Highlights
•Intravital two-photon microscopy shows that TJ remodeling precedes the onset of EAE
•Caveolar transcytosis is not required for endothelial TJ remodeling in vivo
•Effector T cell subsets use distinct mechanisms to cross the inflamed BBB
•Caveolar transcytosis is required for Th1, but not Th17, cell entry into the CNS
Summary
Lymphocytes cross vascular boundaries via either disrupted tight junctions (TJs) or caveolae to induce tissue inflammation. In the CNS, Th17 lymphocytes cross the blood-brain barrier (BBB) before Th1 cells; yet this differential crossing is poorly understood. We have used intravital two-photon imaging of the spinal cord in wild-type and caveolae-deficient mice with fluorescently labeled endothelial tight junctions to determine how tight junction remodeling and caveolae regulate CNS entry of lymphocytes during the experimental autoimmune encephalomyelitis (EAE) model for multiple sclerosis. We find that dynamic tight junction remodeling occurs early in EAE but does not depend upon caveolar transport. Moreover, Th1, but not Th17, lymphocytes are significantly reduced in the inflamed CNS of mice lacking caveolae. Therefore, tight junction remodeling facilitates Th17 migration across the BBB, whereas caveolae promote Th1 entry into the CNS. Moreover, therapies that target both tight junction degradation and caveolar transcytosis may limit lymphocyte infiltration during inflammation.
“Caveolin1 Is Required for Th1 Cell Infiltration, but Not Tight Junction Remodeling, at the Blood-Brain Barrier in Autoimmune Neuroinflammation” by Sarah E. Lutz, Julian R. Smith, Dae Hwan Kim, Carl V.L. Olson, Kyle Ellefsen, Jennifer M. Bates, Sunil P. Gandhi, and Dritan Agalliu in Cell Reports. Published online November 21 2017 doi:10.1016/j.celrep.2017.10.094






