Summary: Researchers use a new technique to image the process of neurotransmission.
Source: University of Maryland School of Medicine.
Study uses cutting-edge technique to image the process of neuronal transmission.
For more than a century, neuroscientists have known that nerve cells talk to one another across the small gaps between them, a process known as synaptic transmission (synapses are the connections between neurons). Information is carried from one cell to the other by neurotransmitters such as glutamate, dopamine, and serotonin, which activate receptors on the receiving neuron to convey excitatory or inhibitory messages.
But beyond this basic outline, the details of how this crucial aspect of brain function occurs have remained elusive. Now, new research by scientists at the University of Maryland School of Medicine (UM SOM) has for the first time elucidated details about the architecture of this process. The paper was published today in the journal Nature.
Synapses are very complicated molecular machines. They are also tiny: only a few millionths of an inch across. They have to be incredibly small, since we need a lot of them; the brain has around 100 trillion of them, and each is individually and precisely tuned to convey stronger or weaker signals between cells.
To visualize features on this sub-microscopic scale, the researchers turned to an innovative technology known as single-molecule imaging, which can locate and track the movement of individual protein molecules within the confines of a single synapse, even in living cells. Using this approach, the scientists identified an unexpected and precise pattern in the process of neurotransmission. The researchers looked at cultured rat synapses, which in terms of overall structure are very similar to human synapses.
“We are seeing things that have never been seen before. This is a totally new area of investigation,” said Thomas Blanpied, PhD, Associate Professor in the Department of Physiology, and leader of the group that performed the work. “For many years, we’ve had a list of the many types of molecules that are found at synapses, but that didn’t get us very far in understanding how these molecules fit together, or how the process really works structurally. Now by using single-molecule imaging to map where many of the key proteins are, we have finally been able to reveal the core architectural structure of the synapse.”
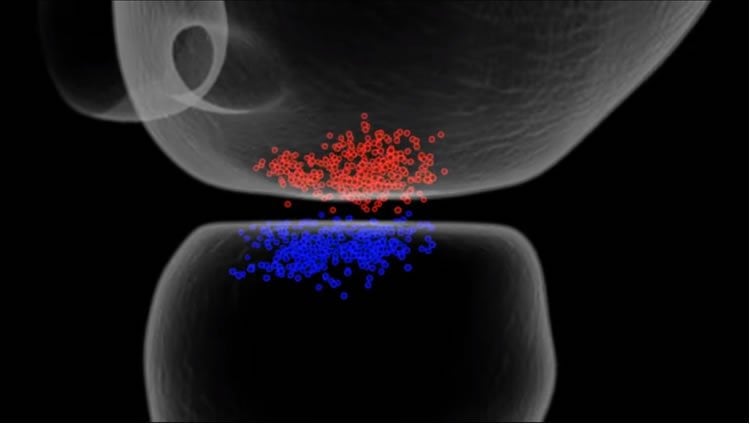
In the paper, Blanpied describes an unexpected aspect to this architecture that may explain why synapses are so efficient, but also susceptible to disruption during disease: at each synapse, key proteins are organized very precisely across the gap between cells. “The neurons do a better job than we ever imagined of positioning the release of neurotransmitter molecules near their receptors,” Blanpied says. “The proteins in the two different neurons are aligned with incredible precision, almost forming a column stretching between the two cells.” This proximity optimizes the power of the transmission, and also suggests new ways that this transmission can be modified.
Understanding this architecture will help clarify how communication within the brain works, or, in the case of psychiatric or neurological disease, how it fails to work. Blanpied is also focusing on the activity of “adhesion molecules,” which stretch from one cell to the other and may be important pieces of the “nano-column.” He suspects that if adhesion molecules are not placed correctly at the synapse, synapse architecture will be disrupted, and neurotransmitters won’t be able to do their jobs. Blanpied hypothesizes that in at least some disorders, the issue may be that even though the brain has the right amount of neurotransmitter, the synapses don’t transmit these molecules efficiently.
Blanpied says that this improved comprehension of synaptic architecture could lead to a better understanding of brain diseases such as depression, schizophrenia and Alzheimer’s disease, and perhaps suggest new ideas for treatments.
Blanpied and his colleagues will next explore whether the synaptic architecture changes in certain disorders: they will begin by looking at a synapses in a mouse model of the pathology in schizophrenia.
Source: David Kohn – University of Maryland School of Medicine
Image Source: This NeuroscienceNews.com image is credited to Jim Stanis.
Video Source: The video is credited to Tom Blanpied.
Original Research: Abstract for “A trans-synaptic nanocolumn aligns neurotransmitter release to receptors” by Ai-Hui Tang, Haiwen Chen, Tuo P. Li, Sarah R. Metzbower, Harold D. MacGillavry and Thomas A. Blanpied in Nature. Published online July 27 2016 doi:10.1038/nature19058
[cbtabs][cbtab title=”MLA”]University of Maryland School of Medicine. “Imaging the Brain Structure That Allows Neurons to Communicate.” NeuroscienceNews. NeuroscienceNews, 27 July 2016.
<https://neurosciencenews.com/neurotransmission-neuroimaging-4744/>.[/cbtab][cbtab title=”APA”]University of Maryland School of Medicine. (2016, July 27). Imaging the Brain Structure That Allows Neurons to Communicate. NeuroscienceNews. Retrieved July 27, 2016 from https://neurosciencenews.com/neurotransmission-neuroimaging-4744/[/cbtab][cbtab title=”Chicago”]University of Maryland School of Medicine. “Imaging the Brain Structure That Allows Neurons to Communicate.” https://neurosciencenews.com/neurotransmission-neuroimaging-4744/ (accessed July 27, 2016).[/cbtab][/cbtabs]
Abstract
A trans-synaptic nanocolumn aligns neurotransmitter release to receptors
Synaptic transmission is maintained by a delicate, sub-synaptic molecular architecture, and even mild alterations in synapse structure drive functional changes during experience-dependent plasticity and pathological disorders. Key to this architecture is how the distribution of presynaptic vesicle fusion sites corresponds to the position of receptors in the postsynaptic density. However, while it has long been recognized that this spatial relationship modulates synaptic strength3, it has not been precisely described, owing in part to the limited resolution of light microscopy. Using localization microscopy, here we show that key proteins mediating vesicle priming and fusion are mutually co-enriched within nanometre-scale subregions of the presynaptic active zone. Through development of a new method to map vesicle fusion positions within single synapses in cultured rat hippocampal neurons, we find that action-potential-evoked fusion is guided by this protein gradient and occurs preferentially in confined areas with higher local density of Rab3-interacting molecule (RIM) within the active zones. These presynaptic RIM nanoclusters closely align with concentrated postsynaptic receptors and scaffolding proteins4, 5, 6, suggesting the existence of a trans-synaptic molecular ‘nanocolumn’. Thus, we propose that the nanoarchitecture of the active zone directs action-potential-evoked vesicle fusion to occur preferentially at sites directly opposing postsynaptic receptor–scaffold ensembles. Remarkably, NMDA receptor activation triggered distinct phases of plasticity in which postsynaptic reorganization was followed by trans-synaptic nanoscale realignment. This architecture suggests a simple organizational principle of central nervous system synapses to maintain and modulate synaptic efficiency.
“A trans-synaptic nanocolumn aligns neurotransmitter release to receptors” by Ai-Hui Tang, Haiwen Chen, Tuo P. Li, Sarah R. Metzbower, Harold D. MacGillavry and Thomas A. Blanpied in Nature. Published online July 27 2016 doi:10.1038/nature19058