Summary: Female mice exposed to a high-fat diet have limited hippocampal neurogenesis. The findings may explain why women are at higher risk of developing dementia than men.
Source: SfN
A high fat diet limits the birth and growth of new neurons in adult female, but not male, mice, according to new research published in eNeuro. Further research could inspire metabolism-based preventions and treatments for brain disorders.
Metabolic disorders like obesity and type 2 diabetes are associated with an increased risk for brain disorders ranging from depression to Alzheimer’s disease. The birth and development of new neurons – adult neurogenesis – may be a link between these two types of conditions. The hippocampus, an area of the brain implicated in memory and emotional processes, is a known site of adult neurogenesis.
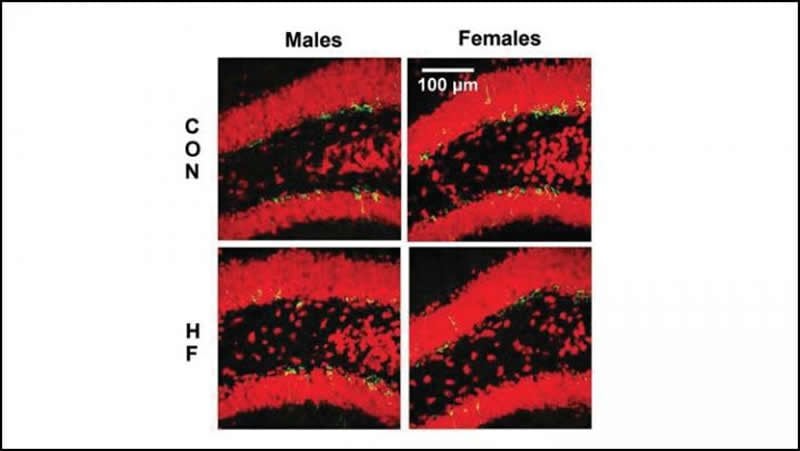
Robison et al. fed one group of mice a high fat diet and another group a normal diet for eighteen weeks. The high fat diet caused weight gain and high blood sugar in both male and female mice, however, only female mice experienced impaired neurogenesis in the hippocampus. The female mice on the high fat diet had fewer newborn and developing neurons, while male mice on the high fat diet had the same number of new neurons as the control mice. This finding offers additional insight into why women are more at risk for greater cognitive decline during Alzheimer’s disease and depression.
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Robison et al., eNeuro 2019.
Original Research: Closed access
“High Fat Diet-Induced Obesity Causes Sex-Specific Deficits in Adult Hippocampal Neurogenesis in Mice”. Lisa S. Robison, Nathan M. Albert, Lauren A. Camargo, Brian M. Anderson, Abigail E. Salinero, David A. Riccio, Charly Abi-Ghanem, Olivia J. Gannon and Kristen L. Zuloaga.
eNeuro doi:10.1523/ENEURO.0391-19.2019.
Abstract
High Fat Diet-Induced Obesity Causes Sex-Specific Deficits in Adult Hippocampal Neurogenesis in Mice
Adult hippocampal neurogenesis (AHN) is suppressed by high fat (HF) diet and metabolic disease, including obesity and Type 2 diabetes. Deficits in AHN may contribute to cognitive decline and increased risk of dementia and mood disorders, which have higher prevalence in women. However, sex differences in the effects of HF diet/metabolic disease on AHN have yet to be thoroughly investigated. Herein, male and female C57BL/6J mice were fed HF or control (CON) diet from ∼2-6 months of age. After 3 months on the diet, mice were injected with EdU, then euthanized 4 weeks later. Cell proliferation, differentiation/maturation and survival of new neurons in the dentate gyrus was assessed with immunofluorescence for EdU, Ki67, doublecortin (DCX), and NeuN. CON females had more proliferating cells (Ki67+) and neuroblasts/immature neurons (DCX+) compared to CON males; however, HF diet reduced these in females to the levels of males. Diet did not affect neurogenesis in males. Further, the numbers of proliferating cells and immature neurons were inversely correlated with both weight gain and glucose intolerance in females only. These effects were robust in the dorsal hippocampus, which supports cognitive processes. Assessment of microglia in the dentate gyrus using immunofluorescence for Iba1 and CD68 uncovered sex-specific effects of diet, which may contribute to observed differences in neurogenesis. These findings demonstrate sex-specific effects of HF diet/metabolic disease on AHN, and highlight the potential for targeting neurogenic deficits to treat cognitive decline and reduce the risk of dementia associated with these conditions, particularly in females.
Significance Statement
Poor diet and metabolic disease, including obesity and Type 2 diabetes, are associated with an increased risk of neurodegenerative and neuropsychiatric disorders, including Alzheimer’s disease, anxiety, and depression. Impaired adult hippocampal neurogenesis may be one mechanism linking these conditions; however, it is unknown whether there are sex-specific effects of high fat diet/metabolic disease on neurogenesis, which could underlie the observed sex difference in these conditions (females more adversely affected than males). We report that high fat diet/metabolic disease impairs cell proliferation and the number of neuroblasts/immature neurons in the dorsal hippocampus, a region that supports cognitive function, in females only. Sex-specific effects of diet on microglia in the subgranular zone were also apparent.






