Summary: Hippocampal neurogenesis continues to occur well into old age, and in those with Alzheimer’s disease. Researchers found evidence of neurogenesis in people up to the age of 99. While neurogenesis continues to occur in those with Alzheimer’s disease, it is significantly reduced compared to those who have normal cognitive function.
Source: University of Illinois
In a new study from the University of Illinois at Chicago, researchers examining post-mortem brain tissue from people ages 79 to 99 found that new neurons continue to form well into old age. The study provides evidence that this occurs even in people with cognitive impairment and Alzheimer’s disease, although neurogenesis is significantly reduced in these people compared to older adults with normal cognitive functioning.
They publish their results in the journal Cell Stem Cell.
The idea that new neurons continue to form into middle age, let alone past adolescence, is controversial, as previous studies have shown conflicting results. The UIC study is the first to find evidence of significant numbers of neural stem cells and newly developing neurons present in the hippocampal tissue of older adults, including those with disorders that affect the hippocampus, which is involved in the formation of memories and in learning.
“We found that there was active neurogenesis in the hippocampus of older adults well into their 90s,” said Orly Lazarov, professor of anatomy and cell biology in the UIC College of Medicine and lead author of the paper.
“The interesting thing is that we also saw some new neurons in the brains of people with Alzheimer’s disease and cognitive impairment.”
She also found that people who scored better on measures of cognitive function had more newly developing neurons in the hippocampus compared to those who scored lower on these tests, regardless of levels of brain pathology.
Lazarov thinks that lower levels of neurogenesis in the hippocampus are associated with symptoms of cognitive decline and reduced synaptic plasticity rather than with the degree of pathology in the brain. For patients with Alzheimer’s disease, pathological hallmarks include deposits of neurotoxic proteins in the brain.
“In brains from people with no cognitive decline who scored well on tests of cognitive function, these people tended to have higher levels of new neural development at the time of their death, regardless of their level of pathology,” Lazarov said. “The mix of the effects of pathology and neurogenesis is complex and we don’t understand exactly how the two interconnect, but there is clearly a lot of variation from individual to individual.”
Lazarov is excited about the therapeutic possibilities of her findings.
“The fact that we found that neural stem cells and new neurons are present in the hippocampus of older adults means that if we can find a way to enhance neurogenesis, through a small molecule, for example, we may be able to slow or prevent cognitive decline in older adults, especially when it starts, which is when interventions can be most effective,” Lazarov said.
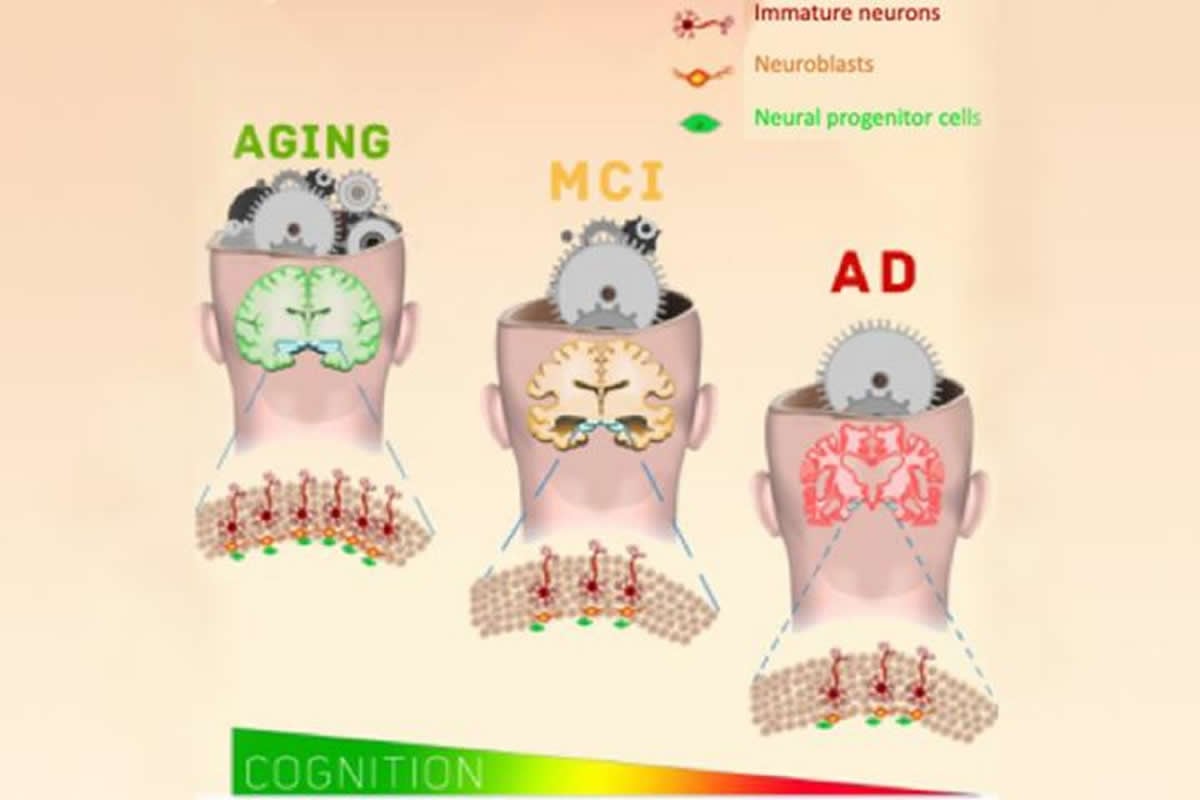
Lazarov and colleagues looked at post-mortem hippocampal tissue from 18 people with an average age of 90.6 years. They stained the tissue for neural stem cells and also for newly developing neurons. They found, on average, approximately 2,000 neural progenitor cells per brain. They also found an average of 150,000 developing neurons. Analysis of a subset of these developing neurons revealed that the number of proliferating developing neurons is significantly lower in people with cognitive impairment and Alzheimer’s disease.
Lazarov is interested in finding out whether the new neurons she and her team discovered in the brains of older adults are behaving the way new neurons do in younger brains.
“There’s still a lot we don’t know about the maturation process of new neurons and the function of neurogenesis in older brains, so it is difficult to predict how much it might ameliorate the effects of cognitive decline and Alzheimer’s disease. The more we find out, the better able we will be to develop interventions that may help preserve cognitive function even in people without Alzheimer’s. We all lose some cognitive function as we age — it’s normal.”
Funding: This research was supported by grants from the National Institute on Aging (AG033570, AG033570-S1, S2, AG060238, AG62251, AG061628, AG17917, AG34374, UH2NS100599) and the Canadian Institutes of Health Research (MT-14037, MOP-81112).
Matthew Tobin, Kianna Musaraca, Ahmed Disouky, Aashutosh Shetti and Abdullah Bheri of UIC; William Honer of the University of British Columbia, Vancouver; and Namhee Kim, Robert Dawe, David Bennett and Konstantinos Arfanakis of Rush University Medical Center are co-authors on the paper.
Source:
University of Illinois
Media Contacts:
Jackie Carey – University of Illinois
Image Source:
The image is credited to Orly Lazarov, et al.
Original Research: Open access
“Human Hippocampal Neurogenesis Persists in Aged Adults and Alzheimer’s Disease Patients”. Orly Lazarov et al.
Cell Stem Cell. doi:10.1016/j.stem.2019.05.003
Abstract
Human Hippocampal Neurogenesis Persists in Aged Adults and Alzheimer’s Disease Patients
Whether hippocampal neurogenesis persists throughout life in the human brain is not fully resolved. Here, we demonstrate that hippocampal neurogenesis is persistent through the tenth decade of life and is detectable in patients with mild cognitive impairments and Alzheimer’s disease. In a cohort of 18 participants with a mean age of 90.6 years, Nestin+Sox2+ neural progenitor cells (NPCs) and DCX+ neuroblasts and immature neurons were detected, but their numbers greatly varied between participants. Nestin+ cells localize in the anterior hippocampus, and NPCs, neuroblasts, and immature neurons are evenly distributed along the anterior to posterior axis. The number of DCX+PCNA+ cells is reduced in mild cognitive impairments, and higher numbers of neuroblasts are associated with better cognitive status. The number of DCX+PCNA+ cells correlates with functional interactions between presynaptic SNARE proteins. Our results suggest that hippocampal neurogenesis persists in the aged and diseased human brain and that it is possibly associated with cognition.






