Summary: Study implicates C4, an immune system gene, in schizophrenia pathogenesis.
Source: PLOS
Excessive activity of an immune system gene previously linked to schizophrenia reproduces neural and behavioral aspects of the disease in mice, according to a new study publishing on January 14 in the open-access journal PLOS Biology by Ashley Comer and Alberto Cruz-Martín of Boston University and colleagues. The finding provides mechanistic support for the importance of the gene in the development of schizophrenia, and may offer a new avenue for therapy development.
Genetics variants that increase the activity of the C4 gene, which encodes part of the immune system’s so-called “complement” cascade, have previously been shown to increase risk for schizophrenia in large genome-wide association studies. However, the mechanism through which such variants might contribute to the emergence of schizophrenia have not been clear because C4 has not previously been experimentally overexpressed.
During normal brain development, it’s known that complement sticks to neuronal synapses and attracts microglia (immune cells in the brain), which engulf synaptic material in the process known as “synaptic pruning.” When mis-regulated, this process can lead to abnormal connectivity in the brain.
Loss of synapses in the prefrontal cortex is a hallmark of schizophrenia, which led the authors to ask whether an overactive C4 gene might contribute to development of schizophrenia through mis-regulation of complement-mediated synaptic pruning by microglia. When they overexpressed complement C4 in the mouse prefrontal cortex, they found an increase in the level of microglial engulfment of synapses, reduced functional connectivity between neurons, and alterations in the properties of neuronal membranes.
Schizophrenia is characterized by deficits in social cognition and social interactions, effects that often precede the development of psychosis. The authors found that juvenile mice with too much complement C4 reduced the time they spent seeking out their mothers compared to control mice, and that adult mice spent less time than control mice interacting with new cage mates.
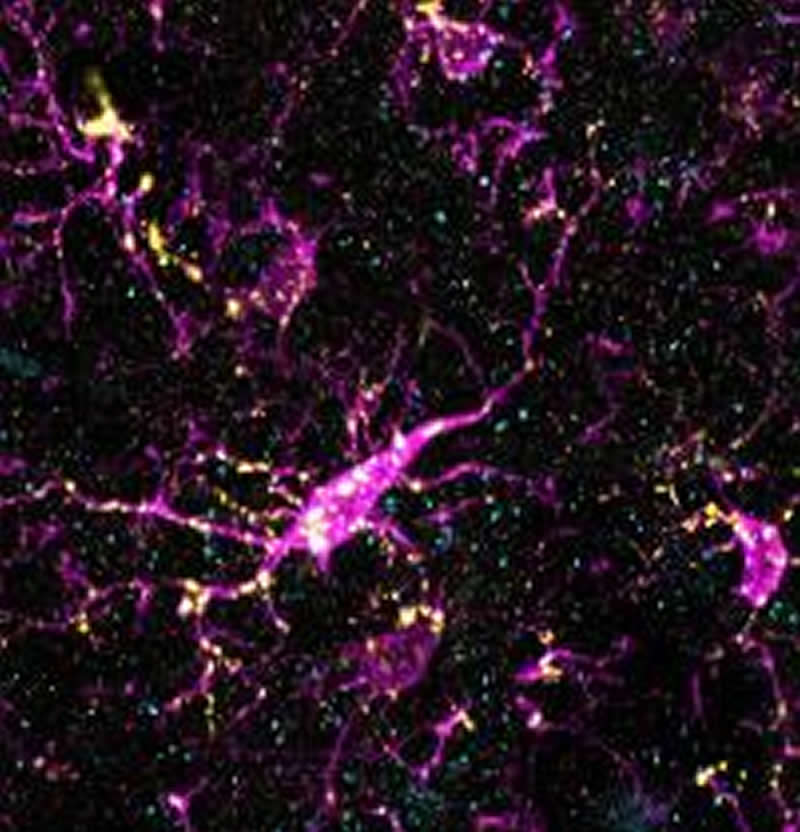
Together, these results suggest that the genetic association found between increased C4 gene activity and schizophrenia may be due at least in part to an inappropriate increase in synaptic pruning during brain development. “This critical developmental window may provide an opportunity to intervene to alter the developmental trajectory of schizophrenia,” Cruz-Martín, the senior investigator, said. While there are currently few approved drugs that can block the complement system, it is an area of intense pharmaceutical research, and these findings are likely to spur even further interest in this approach.
Funding: This work was supported by Biogen (AC-M, #973630), a NARSAD Young Investigator Grant (AC-M, #27202), the Brenton R. Lutz Award (ALC), the NSF NRT UtB: Neurophotonics National Research Fellowship (ALC and LNK, #DGE-1633516), and the Boston University Undergraduate Research Opportunities Program (JL, FSH, TPHN, KLK, ERN, WWY) (https://www.biogen.com/en_us/home.html; https://www.bbrfoundation.org/grants-prizes/grants, https://www.nsf.gov/; http://www.bu.edu/urop/). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing Interests: The authors have declared that no competing interests exist.
Source:
PLOS
Media Contacts:
Alberto Cruz-Martín – PLOS
Image Source:
The image is credited to Rhushikesh A. Phadke.
Original Research: Open access
“Increased expression of schizophrenia-associated gene C4 leads to hypoconnectivity of prefrontal cortex and reduced social interaction”. Rhushikesh A. Phadke et al.
PLOS Biology doi:10.1371/journal.pbio.3000604.
Abstract
Increased expression of schizophrenia-associated gene C4 leads to hypoconnectivity of prefrontal cortex and reduced social interaction
Schizophrenia is a severe mental disorder with an unclear pathophysiology. Increased expression of the immune gene C4 has been linked to a greater risk of developing schizophrenia; however, it is not known whether C4 plays a causative role in this brain disorder. Using confocal imaging and whole-cell electrophysiology, we demonstrate that overexpression of C4 in mouse prefrontal cortex neurons leads to perturbations in dendritic spine development and hypoconnectivity, which mirror neuropathologies found in schizophrenia patients. We find evidence that microglia-mediated synaptic engulfment is enhanced with increased expression of C4. We also show that C4-dependent circuit dysfunction in the frontal cortex leads to decreased social interactions in juvenile and adult mice. These results demonstrate that increased expression of the schizophrenia-associated gene C4 causes aberrant circuit wiring in the developing prefrontal cortex and leads to deficits in juvenile and adult social behavior, suggesting that altered C4 expression contributes directly to schizophrenia pathogenesis.






