A study by UT Southwestern Medical Center researchers published online today in Nature reveals new insight into why the most common, deadly kind of brain tumor in adults recurs and identifies a potential target for future therapies.
Glioblastoma multiforme (GBM) currently is considered incurable. Despite responding to initial therapy, the cancer almost always returns. GBM is a fast-growing, malignant brain tumor that occurred in 15 percent of the estimated 22,000 Americans diagnosed with brain and nervous system tumors in 2010. The median survival rate is about 15 months, according to the National Cancer Institute.
“We identified a subset of brain tumor cells that are slower growing or remain at rest, and appear to be the source of cancer recurrence after standard therapy in which the drug temozolomide is given to stop the tumor’s growth,” said Dr. Luis Parada, chairman of developmental biology and director of the Kent Waldrep Center for Basic Research on Nerve Growth and Regeneration. “Current therapy targets fast-growing tumor cells but not those responsible for new tumors. To the best of our knowledge, this is the first identification of a cancer stem-like cell in a spontaneously forming tumor inside a mammal.”
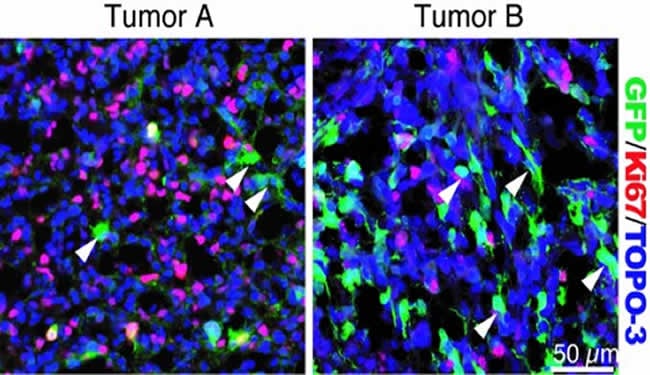
Using a genetically engineered mouse model of GBM, the researchers found that the resting tumor cells act more like stem cells – the non-cancerous cells the body uses to repair and replenish itself – which exist in a resting state until needed, he explained.
The existence of cancer stem cells in solid tumors remains controversial, Dr. Parada said, with some scientists in the field taking the concept for granted and others rejecting it outright. In addition, the definition of a cancer stem cell is a moving target, hence the use of the term stem-like cell in this study, he said.
“We are trying to better understand these cells. The important point is that we now are faced with technical obstacles, not conceptual ones,” said Dr. Parada.
Notes about this brain cancer research
Other UT Southwestern researchers involved include lead author and former postdoctoral student of developmental biology Dr. Jian Chen, who is now a senior scientist at OriGene Technologies in Wuxi, China; Yanjiao Li, a research associate of developmental biology; Dr. Tzong-Schiue Yu, a former graduate student of developmental biology and pediatrics; Dr. Renée M. McKay, assistant professor of developmental biology; Dr. Dennis K. Burns, professor of pathology; and Dr. Steven G. Kernie, a former associate professor of pediatrics and developmental biology. Drs. Kernie and Yu are now at Columbia University Medical Center.
Funding: The study was supported by grants from the National Institutes of Health, the Cancer Prevention and Research Institute of Texas, the Goldhirsh Foundation, and the James S. McDonnell Foundation.
Contact: Deborah Wormser – The University of Texas Southwestern Medical Center
Source: The University of Texas Southwestern Medical Center press release
Image Source: Neuroscience image adapted from press release image and provided courtesy of Nature
Original Research: Abstract for “A restricted cell population propagates glioblastoma growth after chemotherapy” by Jian Chen, Yanjiao Li, Tzong-Shiue Yu, Renée M. McKay, Dennis K. Burns, Steven G. Kernie and Luis F. Parada in Nature online 1 August 2012 doi:10.1038/nature11287