Summary: A new study suggests developing new therapeutics that use cannabinoid receptors to treat chronic pain.
Source: OHSU.
Journal of Neuroscience study examines medicinal properties of cannabis.
OHSU research suggests an avenue for developing treatments for chronic pain that harness the medicinal properties of cannabis while minimizing the threat of addiction.
The study, conducted in a rodent model, provides additional rationale for the development of therapeutics using cannabinoid receptors to treat chronic pain, which afflicts about 30 percent of the U.S. population. OHSU investigators studied the function of two forms of cell membrane receptors that bind cannabinoids that occur naturally within the body, called endocannabinoids.
“It may be an avenue where we can get better pain medications that are not addictive,” said senior author Susan Ingram, Ph.D., an associate professor of neurosurgery in the OHSU School of Medicine.
Ingram and colleagues report the treatment of chronic pain has challenged the medical system, with medications that are ineffective or create serious side effects: “However, emerging data indicate that drugs that target the endocannabinoid system might produce analgesia with fewer side effects compared with opioids.”
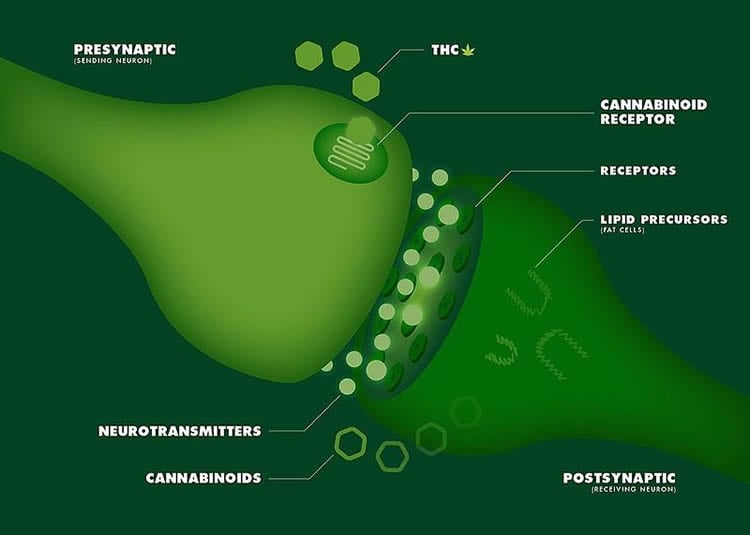
The body’s endocannabinoid system comprises receptors, endocannabinoid molecules and enzymes that make and degrade the endocannabinoids located in the brain and throughout the central and peripheral nervous system. The research team focused on two cannabinoid receptors, known as CB1 and CB2, in the rostral ventromedial medulla – a group of neurons located in the brainstem known to modulate pain. The study is the first to examine CB1 and CB2 receptor function at the membrane level in late adolescent and adult neurons.
The researchers observed that chronic inflammatory pain increased activity of CB2 receptors and decreased CB1 activity. Cannabis activates both CB1 and CB2 receptors equally. The study suggests that selective activation of CB2 receptors contributes to the medicinal benefit of cannabis while minimizing the propensity of the other cannabinoid receptor, CB1, to induce tolerance and withdrawal. Ingram said the next phase of the research will further explore this area of brain circuitry, which ultimately could lead to the development of a new class of pain medications.
Co-authors include lead author Ming-Hua Li, Ph.D., and Katherine L. Suchland, both with the Department of Neurological Surgery, OHSU School of Medicine.
Funding: The study was funded by grants from the National Institutes of Health (DA035316 and R56NS093894) and American Heart Association (13SDG14590005, MH.L.).
Source: Erik Robinson – OHSU
Image Source: NeuroscienceNews.com image is credited to VICE media and is licensed CC BY SA 4.0.
Original Research: Abstract for “Compensatory activation of cannabinoid CB2 receptor inhibition of GABA release in the rostral ventromedial medulla (RVM) in inflammatory pain” by Ming-Hua Li, Katherine L. Suchland and Susan L. Ingram in Journal of Neuroscience. Published online December 9 2016 doi:10.1523/JNEUROSCI.1310-16.2016
[cbtabs][cbtab title=”MLA”]OHSU “Harnessing Pain Relieving Properties of Cannabis Without Addictive Effects.” NeuroscienceNews. NeuroscienceNews, 12 January 2017.
<https://neurosciencenews.com/pain-relief-neurology-5928/>.[/cbtab][cbtab title=”APA”]OHSU (2017, January 12). Harnessing Pain Relieving Properties of Cannabis Without Addictive Effects. NeuroscienceNew. Retrieved January 12, 2017 from https://neurosciencenews.com/pain-relief-neurology-5928/[/cbtab][cbtab title=”Chicago”]OHSU “Harnessing Pain Relieving Properties of Cannabis Without Addictive Effects.” https://neurosciencenews.com/pain-relief-neurology-5928/ (accessed January 12, 2017).[/cbtab][/cbtabs]
Abstract
Compensatory activation of cannabinoid CB2 receptor inhibition of GABA release in the rostral ventromedial medulla (RVM) in inflammatory pain
The rostral ventromedial medulla (RVM) is a relay in the descending pain modulatory system and an important site of endocannabinoid modulation of pain. Endocannabinoids inhibit GABA release in the RVM but it is not known if this effect persists in chronic pain states. In the present studies, persistent inflammation induced by complete Freund’s adjuvant (CFA) increased GABAergic miniature inhibitory postsynaptic currents (mIPSCs). Endocannabinoid activation of cannabinoid (CB1) receptors known to inhibit presynaptic GABA release was significantly reduced in the RVM of CFA-treated rats compared to naïve rats. The reduction in CFA-treated rats correlated with decreased CB1 receptor protein expression and function in the RVM. Paradoxically, the non-selective CB1/CB2 receptor agonist WIN55,212 inhibited GABAergic mIPSCs in both naïve and CFA-treated rats. However, WIN55,212 inhibition was reversed by the CB1 receptor antagonist rimonabant in naïve rats but not in CFA-treated rats. WIN55,212-mediated inhibition in CFA-treated rats was blocked by the CB2 receptor-selective antagonist SR144528 indicating that CB2 receptor function in the RVM is increased during persistent inflammation. Consistent with these results, CB2 receptor agonists, AM1241 and GW405833 inhibited GABAergic mIPSC frequency only in CFA-treated rats and the inhibition was reversed with SR144258. When administered alone, SR144528 and another CB2 receptor-selective antagonist AM630 increased mIPSC frequency in the RVM of CFA-treated rats indicating that CB2 receptors are tonically activated by endocannabinoids. Our data provide evidence that CB2 receptor function emerges in the RVM in persistent inflammation and that selective CB2 receptor agonists may be useful for treatment of persistent inflammatory pain.
SIGNIFICANCE STATEMENT
These studies demonstrate that endocannabinoid signaling to CB1- and CB2-receptors in adult RVM is altered in persistent inflammation. The emergence of CB2 receptor function in the RVM provides additional rationale for the development of CB2 receptor-selective agonists as useful therapeutics for chronic inflammatory pain.
“Compensatory activation of cannabinoid CB2 receptor inhibition of GABA release in the rostral ventromedial medulla (RVM) in inflammatory pain” by Ming-Hua Li, Katherine L. Suchland and Susan L. Ingram in Journal of Neuroscience. Published online December 9 2016 doi:10.1523/JNEUROSCI.1310-16.2016






