Summary: A new study reports high contrast visual stimulation can help damaged retinal neurons to regrow optic nerve fibers.
Source: NIH/NEI.
NIH-funded mouse study is the first to show that visual stimulation helps re-wire the visual system and partially restores sight.
A study in mice funded by the National Institutes of Health (NIH) shows for the first time that high-contrast visual stimulation can help damaged retinal neurons regrow optic nerve fibers, otherwise known as retinal ganglion cell axons. In combination with chemically induced neural stimulation, axons grew further than in strategies tried previously. Treated mice partially regained visual function. The study also demonstrates that adult regenerated central nervous system (CNS) axons are capable of navigating to correct targets in the brain. The research was funded through the National Eye Institute (NEI), a part of NIH.
“Reconnecting neurons in the visual system is one of the biggest challenges to developing regenerative therapies for blinding eye diseases like glaucoma,” said NEI Director Paul A. Sieving, M.D., Ph.D. “This research shows that mammals have a greater capacity for central nervous system regeneration than previously known.”
The optic nerve is the eye’s data cable, carrying visual information from the light-sensing neurons of the retina to the brain. Like a bundle of wires, it consists of about a million axons that each extend from an individual retinal ganglion cell. A variety of optic neuropathies, such as glaucoma, cause vision loss when they destroy or damage these axons. In adults, retinal ganglion cell axons fail to regrow on their own, which is why vision loss from optic neuropathies is usually permanent.
The researchers induced optic nerve damage in mice using forceps to crush the optic nerve of one eye just behind the eyeball. The mice were then placed in a chamber several hours a day for three weeks where they viewed high-contrast images–essentially changing patterns of black lines. The mice had modest but significant axonal regrowth compared to control mice that did not receive the high-contrast visual stimulation.
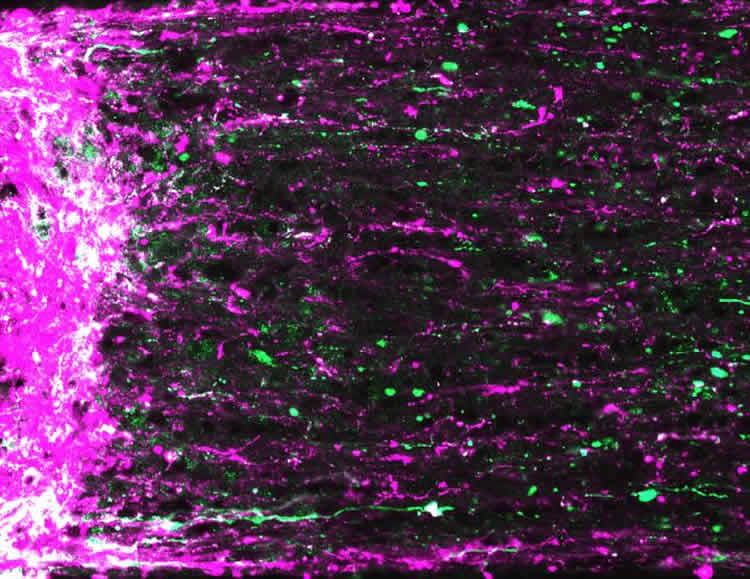
Prior work by the scientists showed that increasing activity of protein called mTOR promoted optic nerve regeneration. And so they wondered if combining visual stimulation with increased mTOR activity might have a synergistic effect. Two weeks prior to nerve crush, the scientists used gene therapy to cause the retinal ganglion cells to overexpress mTOR. Optic nerve crush was performed and mice were exposed to high-contrast visual stimulation daily. After three weeks, the scientists saw more extensive regeneration, with axons growing through the optic nerve as far as the optic chiasm, a distance from the eye of about 6 millimeters. Encouraged by these results, the researchers again increased mTOR activity but then forced mice to use the treated eye during visual stimulation by suturing shut the good eye. This combined approach of increasing mTOR activity with intense visual stimulation promoted regeneration down the full length of the optic nerve and into various visual centers of the brain.
“We saw the most remarkable growth when we closed the good eye, forcing the mice to look through the injured eye,” said Andrew Huberman, Ph.D., associate professor, Stanford University School of Medicine’s department of neurobiology, and lead author of the report, published online today in Nature Neuroscience. In three weeks, the axons grew as much as 12 millimeters, a rate about 500 times faster than untreated CNS axons.
The regenerating axons also navigated to the correct brain regions, a finding that Huberman said sheds light on a pivotal question in regenerative medicine: “If a nerve cell can regenerate, does it wander or does it recapitulate its developmental program and find its way back to the correct brain areas?”
Using transgenic mouse lines designed to express fluorescent proteins only in specific retinal ganglion cell subtypes (about 30 exist), the investigators traced where regenerating axons went. “The two types of retinal ganglion cells that we looked at–α-cells and melanopsin cells–seemed fully capable of navigating back to correct locations in the brain, plugging in and forming synapses,” said Huberman. “And just as interesting, they didn’t go to the wrong places.” Fluorescent axons appeared in brain regions where α-cells and melanopsin cells would be expected but were absent in other regions.
Visual function was partially restored in animals that received visual/mTOR combination therapy. The investigators used four tests to assess four types of visual perception: ability to track moving objects, pupillary reflex, depth perception, and ability to detect an overhead predator–a stimulus that normally causes mice to freeze or flee for cover. Mice treated with combination therapy performed significantly better than untreated mice in two of the four tests.
“This study’s striking finding that activity promotes nerve regrowth holds great promise for therapies aimed at degenerative retinal diseases,” noted Thomas Greenwell, NEI program director for retinal neuroscience research. Greenwell said the research has great relevance to the NEI Audacious Goals Initiative (AGI), a sustained effort to develop regenerative medicine for retinal diseases.
For future therapies that preserve optic nerve axons, Huberman envisions the development of filters for virtual reality video games, television programs, or eyeglasses designed to deliver regeneration-inducing visual stimulation. A drawback of the optic nerve crush model is that it does not mimic typical blinding diseases or injuries. The investigators are therefore currently examining the effect of intense visual stimulation in a mouse glaucoma model. Going forward, they are homing in on the specific qualities of visual stimulation that drive retinal regeneration.
Funding: This research was funded by NIH grant EY026100 and the Glaucoma Research Foundation.
Source: Dustin Hays – NIH/NEI
Image Source: This NeuroscienceNews.com image is credited to Andrew D. Huberman.
Original Research: The study will appear in Nature Neuroscience during the week of July 11 2016.
[cbtabs][cbtab title=”MLA”]NIH/NEI. “Visual Activity Regenerates Neural Connections Between the Eye and Brain.” NeuroscienceNews. NeuroscienceNews, 11 July 2016.
<https://neurosciencenews.com/neuroregeneration-brain-eye-vision-4652/>.[/cbtab][cbtab title=”APA”]NIH/NEI. (2016, July 11). Visual Activity Regenerates Neural Connections Between the Eye and Brain. NeuroscienceNews. Retrieved July 11, 2016 from https://neurosciencenews.com/neuroregeneration-brain-eye-vision-4652/[/cbtab][cbtab title=”Chicago”]NIH/NEI. “Visual Activity Regenerates Neural Connections Between the Eye and Brain.” https://neurosciencenews.com/neuroregeneration-brain-eye-vision-4652/ (accessed July 11, 2016).[/cbtab][/cbtabs]







