Summary: Dentate gyrus neurogenesis acts to replace lost neurons and restore function following massive neuron loss.
Source: SfN
The ability to create new neurons may exist as built-in protection for sensitive brain areas, according to research in mice recently published in Journal of Neuroscience.
For a quarter of a century, scientists have known that the brain creates new neurons even into adulthood – a process called adult neurogenesis. The question has been: why? Adult neurogenesis occurs in the dentate gyrus, a brain region involved in memory, but which is also susceptible to damage in the early stages of epilepsy and Alzheimer’s disease. Much research has focused on how adult neurogenesis affects learning and memory, but new research suggests an additional role: regeneration.
Licht et al. tested this by exposing the brains of mice to a high level of VEGF, a blood vessel growth protein that can become toxic to the dentate gyrus, for three months. The team then monitored how the dentate gyrus recovered. Within a few months, it had almost fully repaired itself through the activity of adult neural stem cells. The restored dentate gyri displayed similar size, connectivity, and activity to unharmed dentate gyri.
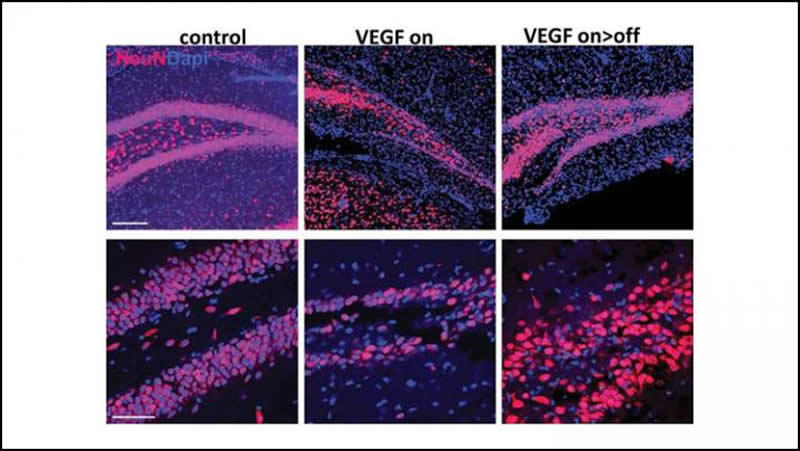
The regenerative function of neurogenesis suggests the process may exist as a built-in brain repair system, akin to stem cells in skin and bone marrow.
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Licht et al., JNeurosci 2020.
Original Research: Closed access
“Age-Dependent Remarkable Regenerative Potential of the Dentate Gyrus Provided by Intrinsic Stem Cells”. Tamar Licht, Tirzah Kreisel, Yoav Biala, Sandesh Mohan, Yoel Yaari, Andrey Anisimov, Kari Alitalo and Eli Keshet.
Journal of Neuroscience doi:10.1523/JNEUROSCI.1010-19.2019.
Abstract
Age-Dependent Remarkable Regenerative Potential of the Dentate Gyrus Provided by Intrinsic Stem Cells
Multiple insults to the brain lead to neuronal cell death, thus raising the question to what extent can lost neurons be replenished by adult neurogenesis. Here we focused on the hippocampus and especially the dentate gyrus (DG), a vulnerable brain region and one of the two sites where adult neuronal stem cells (NSCs) reside. While adult hippocampal neurogenesis was extensively studied with regard to its contribution to cognitive enhancement, we focused on their underestimated capability to repair a massively injured, nonfunctional DG. To address this issue, we inflicted substantial DG-specific damage in mice of either sex either by diphtheria toxin-based ablation of >50% of mature DG granule cells (GCs) or by prolonged brain-specific VEGF overexpression culminating in extensive, highly selective loss of DG GCs (thereby also reinforcing the notion of selective DG vulnerability). The neurogenic system promoted effective regeneration by increasing NSCs proliferation/survival rates, restoring a nearly original DG mass, promoting proper rewiring of regenerated neurons to their afferent and efferent partners and regaining of lost spatial memory. Notably, concomitantly with the natural age-related decline in the levels of neurogenesis, the regenerative capacity of the hippocampus also subsided with age. The study thus revealed an unappreciated regenerative potential of the young DG and suggests hippocampal NSCs as a critical reservoir enabling recovery from catastrophic DG damage.
SIGNIFICANCE STATEMENT
Adult hippocampal neurogenesis has been extensively studied in the context of its role in cognitive enhancement but whether, and to what extent can dentate gyrus (DG)-resident neural stem cells drive regeneration of an injured DG has remained unclear. Here we show that DG neurogenesis act to replace lost neurons and restore lost functions even following massive (>50%) neuronal loss. Age-related decline of neurogenesis is paralleled by a progressive decline of regenerative capacity. Considering also the exceptional vulnerability of the DG to insults, these findings provide a further rationale for maintaining DG neurogenesis in adult life.






