The death of brain cells in Parkinson’s disease may be caused by a form of cellular energy crisis in neurons that require unusually high quantities of energy to carry out their job of regulating movement, researchers at the University of Montreal reported today. The neurodegenerative disorder affects over 100,000 Canadians and over 1,000,000 Americans. “Like a motor constantly running at high speed, these neurons need to produce an incredible amount of energy to function. They appear to exhaust themselves and die prematurely,” said lead researcher Louis-Éric Trudeau, a professor at the university’s Departments of Phamacology and Neurosciences. The research article, published today in Current Biology, includes contributions from Consiglia Pacelli, Nicolas Giguère and Marie-Josée Bourque, also of the University of Montreal, and Martin Lévesque and Ruth Slack, of Laval and Ottawa universities, respectively.
The findings are in some ways a culmination of Trudeau’s 17 years of studying the part of the brain that causes Parkinson’s disease, schizophrenia and drug addiction. His findings could open the door to the creation of better animal models of Parkinson’s disease and the identification of new treatment strategies. “For some unknown reason, it has been incredibly difficult to reproduce the symptoms of Parkinson’s in mice, even when introducing in the genome of these animals the same mutations found in humans afflicted by familial forms of the disease. Our discovery provides a new lead to potentially overcome such difficulties” Trudeau explained.
Improved animal models open a variety of new avenues of research. “It’s possible that new medications could be developed to help the neurons in question reduce their energy consumption or produce energy more efficiently, which would reduce accumulated damage over the years,” Trudeau said. His team is already looking at the possible next steps with Professor Slack and her colleague Professor David Park.
Targeting the dark side
Unlike Alzheimer’s, which has a wider-ranging impact on billions of brain neurons, the primary symptoms or Parkinson’s are caused by the death of tens or hundreds of thousands of neurons in a few more restricted areas of the brain, including regions called the substantia nigra (literally “the black substance”), the locus ceruleus and the dorsal nucleus of the vagus nerve.
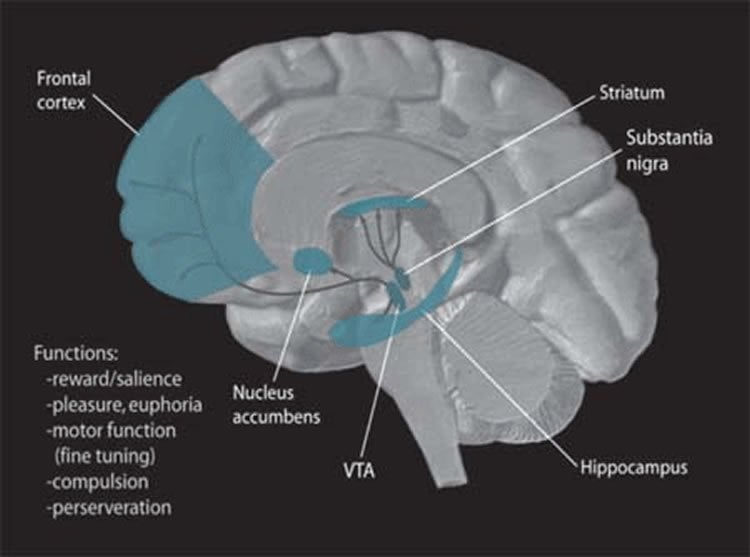
Key to the mystery may be mitochondria, the powerhouses that allow cells to grow and neurons to conduct electrical signals and release their chemical messengers such as dopamine, noradrenaline and acetylcholine. For the past three years, the research team carried out numerous experiments in order to identify why mitochondria in neurons of the substantia nigra work so hard and apparently lead neurons to “overheat”.
They discovered that this overheating could be caused by the fact that these neurons have an amazingly complex structure with a large number of extensions and neurotransmitter release sites, much like a tree with numerous branches. Providing energy to these numerous branches may make the neurons particularly vulnerable, leading, in the context of aging, to malfunction and cell death, thus triggering Parkinson’s, with the onset of symptoms generally at around age sixty. “Our work supports the theory that very complex neurons like those found in the substantia nigra force the mitochondria to constantly work at burnout rates to produce energy. This would explain the accelerated cell deterioration,” Trudeau explained. “To use the analogy of a motor, a car that overheats will burn significantly more fuel, and, not surprisingly, end up at the garage more often.”
Professor Trudeau notes that the most common neurodegenerative diseases are particularly challenging for researchers, because in a way, their increasing prevalence is the result of increased life expectancy. “From an evolutionary standpoint, some of our neurons are perhaps just not programmed to last 80, 90 or 100 years, as we are seeing more and more. It’s to be expected that certain parts of our body are less able to withstand the effects of time,” he said. However, given the more localized nature of Parkinson’s disease (compared to other afflictions), an effective treatment may be discovered in the not-too-distant future. Nevertheless, Trudeau points out that his primary goal is to develop a fundamental understanding of the mechanisms of the brain in order to shed new light on neurological disorders.
Professor Louis-Éric Trudeau and his team are affiliated with the University of Montreal’s Groupe de Recherche sur le Système Nerveux Central, Department of Pharmacology and Department of Neurosciences.
Funding: The research team received support for this project from Brain Canada in partnership with the Krembil Foundation, as well as from Parkinson Society Canada.
Source: William Raillant-Clark – University of Montreal
Image Source: The image is in the public domain
Original Research: Abstract for “Elevated mitochondrial bioenergetics and axonal arborization size are key contributors to the vulnerability of dopamine neurons” by Consiglia Pacelli, Nicolas Giguère, Marie-Josée Bourque, Martin Lévesque, Ruth S. Slack, and Louis-Éric Trudeau in Current Biology. Published online August 27 2015 doi:10.1016/j.cub.2015.07.050
Abstract
Elevated mitochondrial bioenergetics and axonal arborization size are key contributors to the vulnerability of dopamine neurons
Highlights
•Vulnerable SNc DA neurons differ in many ways from less vulnerable VTA DA neurons
•SNc DA neurons have a higher basal rate of oxidative phosphorylation (OXPHOS)
•SNc DA neurons have a more complex axon and a higher density of axonal mitochondria
•Reducing axonal arborization size and basal OXPHOS decreases oxidative stress and vulnerability
Summary
Although the mechanisms underlying the loss of neurons in Parkinson’s disease are not well understood, impaired mitochondrial function and pathological protein aggregation are suspected as playing a major role. Why DA (dopamine) neurons and a select small subset of brain nuclei are particularly vulnerable to such ubiquitous cellular dysfunctions is presently one of the key unanswered questions in Parkinson’s disease research. One intriguing hypothesis is that their heightened vulnerability is a consequence of their elevated bioenergetic requirements. Here, we show for the first time that vulnerable nigral DA neurons differ from less vulnerable DA neurons such as those of the VTA (ventral tegmental area) by having a higher basal rate of mitochondrial OXPHOS (oxidative phosphorylation), a smaller reserve capacity, a higher density of axonal mitochondria, an elevated level of basal oxidative stress, and a considerably more complex axonal arborization. Furthermore, we demonstrate that reducing axonal arborization by acting on axon guidance pathways with Semaphorin 7A reduces in parallel the basal rate of mitochondrial OXPHOS and the vulnerability of nigral DA neurons to the neurotoxic agents MPP+ (1-methyl-4-phenylpyridinium) and rotenone. Blocking L-type calcium channels with isradipine was protective against MPP+ but not rotenone. Our data provide the most direct demonstration to date in favor of the hypothesis that the heightened vulnerability of nigral DA neurons in Parkinson’s disease is directly due to their particular bioenergetic and morphological characteristics.
“Activating positive memory engrams suppresses depression-like behaviour” by Steve Ramirez, Xu Liu, Christopher J. MacDonald, Anthony Moffa, Joanne Zhou, Roger L. Redondo and Susumu Tonegawa in Nature. Published online June 18 2015 doi:10.1038/nature14514







