A new study out today in the journal Nature Communications shows that cells normally associated with protecting the brain from infection and injury also play an important role in rewiring the connections between nerve cells. While this discovery sheds new light on the mechanics of neuroplasticity, it could also help explain diseases like autism spectrum disorders, schizophrenia, and dementia, which may arise when this process breaks down and connections between brain cells are not formed or removed correctly.
“We have long considered the reorganization of the brain’s network of connections as solely the domain of neurons,” said Ania Majewska, Ph.D., an associate professor in the Department of Neuroscience at the University of Rochester Medical Center (URMC) and senior author of the study.
“These findings show that a precisely choreographed interaction between multiple cells types is necessary to carry out the formation and destruction of connections that allow proper signaling in the brain.”
The study is another example of a dramatic shift in scientists’ understanding of the role that the immune system, specifically cells called microglia, plays in maintaining brain function. Microglia have been long understood to be the sentinels of the central nervous system, patrolling the brain and spinal cord and springing into action to stamp out infections or gobble up dead cell tissue. However, scientists are now beginning to appreciate that, in addition to serving as the brain’s first line of defense, these cells also have a nurturing side, particularly as it relates to the connections between neurons.
The formation and removal of the physical connections between neurons is a critical part of maintaining a healthy brain and the process of creating new pathways and networks among brain cells enables us to absorb, learn, and memorize new information.
“The brain’s network of connections is like a garden,” said Rebecca Lowery, a graduate student in Majewska’s lab and co-author of the study. “Not only does it require nourishment and a healthy environment, but every once in a while you need to prune dead branches and pull up weeds in order to allow new flowers to grow.”
While this constant reorganization of neural networks – called neuroplasticity – has been well understood for some time, the basic mechanisms by which connections between brain cells are made and broken has eluded scientists.
Performing experiments in mice, the researchers employed a well-established model of measuring neuroplasticity by observing how cells reorganize their connections when visual information received by the brain is reduced from two eyes to one.
The researchers found that in the mice’s brains microglia responded rapidly to changes in neuronal activity as the brain adapted to processing information from only one eye. They observed that the microglia targeted the synaptic cleft – the business end of the connection that transmits signals between neurons. The microglia “pulled up” the appropriate connections, physically disconnecting one neuron from another, while leaving other important connections intact.
This is similar to what occurs during an infection or injury, in which microglia are activated, quickly navigate towards the injured site, and remove dead or diseased tissue while leaving healthy tissue untouched.
The researchers also pinpointed one of the key molecular mechanisms in this process and observed that when a single receptor – called P2Y12 – was turned off the microglia ceased removing the connections between neurons.
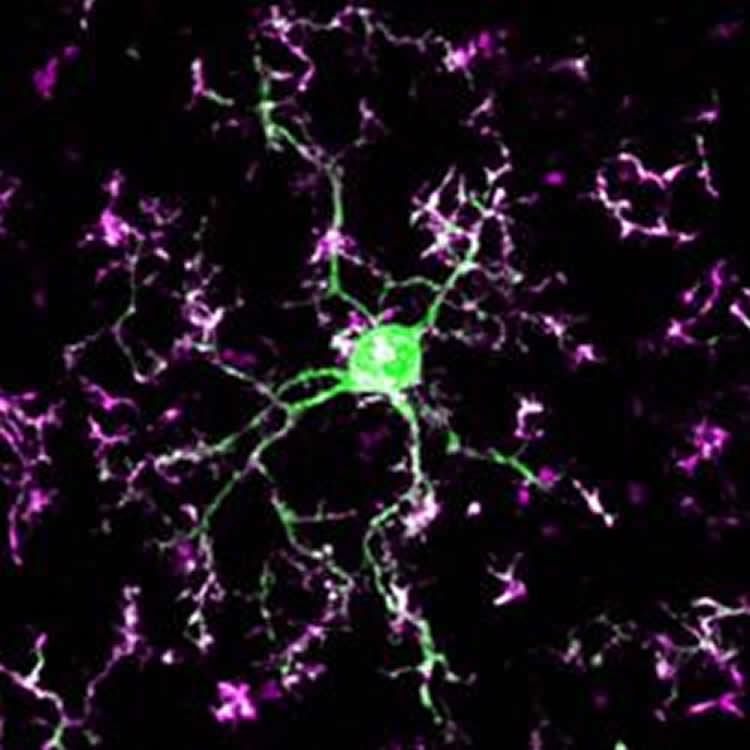
These findings may provide new insight into disorders that are the characterized by sensory or cognitive dysfunction, such as autism spectrum disorders, schizophrenia, and dementia. It is possible that when the microglia’s synapse pruning function is interrupted or when the cells mistakenly remove the wrong connections – perhaps due to genetic factors or because the cells are too occupied elsewhere fighting an infection or injury – the result is impaired signaling between brain cells.
“These findings demonstrate that microglia are a dynamic and integral component of the complex machinery that allows neurons to reorganize their connections in the healthy mature brain,” said Grayson Sipe, a graduate student in Majewska’s lab and co-author of the study. “While more work needs to be done to fully understand this process, this study may help us understand how genetics or disruption of the immune system contributes to neurological disorders.”
Additional co-authors include Emily Kelly and Cassandra Lamantia with URMC and Marie Eve Tremblay with Laval University in Quebec.
Funding: The study was support by the National Eye Institute and the National Institute for Neurological Disorders and Stroke.
Source: Mark Michaud – University of Rochester
Image Source: The image is adapted from the University of Rochester press release.
Original Research: Full open access research for “Microglial P2Y12 is necessary for synaptic plasticity in mouse visual cortex” by G. O. Sipe, R. L. Lowery,, M-È Tremblay, E. A. Kelly, C. E. Lamantia and A. K. Majewska in Nature Communications. Published online March 7 2016 doi:10.1038/ncomms10905
Abstract
Microglial P2Y12 is necessary for synaptic plasticity in mouse visual cortex
Microglia are the resident immune cells of the brain. Increasingly, they are recognized as important mediators of normal neurophysiology, particularly during early development. Here we demonstrate that microglia are critical for ocular dominance plasticity. During the visual critical period, closure of one eye elicits changes in the structure and function of connections underlying binocular responses of neurons in the visual cortex. We find that microglia respond to monocular deprivation during the critical period, altering their morphology, motility and phagocytic behaviour as well as interactions with synapses. To explore the underlying mechanism, we focused on the P2Y12 purinergic receptor, which is selectively expressed in non-activated microglia and mediates process motility during early injury responses. We find that disrupting this receptor alters the microglial response to monocular deprivation and abrogates ocular dominance plasticity. These results suggest that microglia actively contribute to experience-dependent plasticity in the adolescent brain.
“Microglial P2Y12 is necessary for synaptic plasticity in mouse visual cortex” by G. O. Sipe, R. L. Lowery,, M-È Tremblay, E. A. Kelly, C. E. Lamantia and A. K. Majewska in Nature Communications. Published online March 7 2016 doi:10.1038/ncomms10905






