Summary: Learning how to manipulate inflammatory conditions could help to develop new therapies to stimulate neurogenesis and protect against Alzheimer’s, a new study suggests.
Source: DZNE.
Study suggests zebrafish might teach us how to tackle Alzheimer’s disease.
Zebrafish and humans are obviously quite different species. However, they share an evolutionary past that translates into various similarities on the cellular and molecular scale. “The regenerative skills apparent in zebrafish might lie dormant in humans and might somehow be activated by pulling the right strings”, Dr. Kizil explains. “This is why we study if and how zebrafish cope with neurodegeneration. We want to understand the basic molecular mechanisms of such a regenerative aptitude in order to design better clinical therapies.”
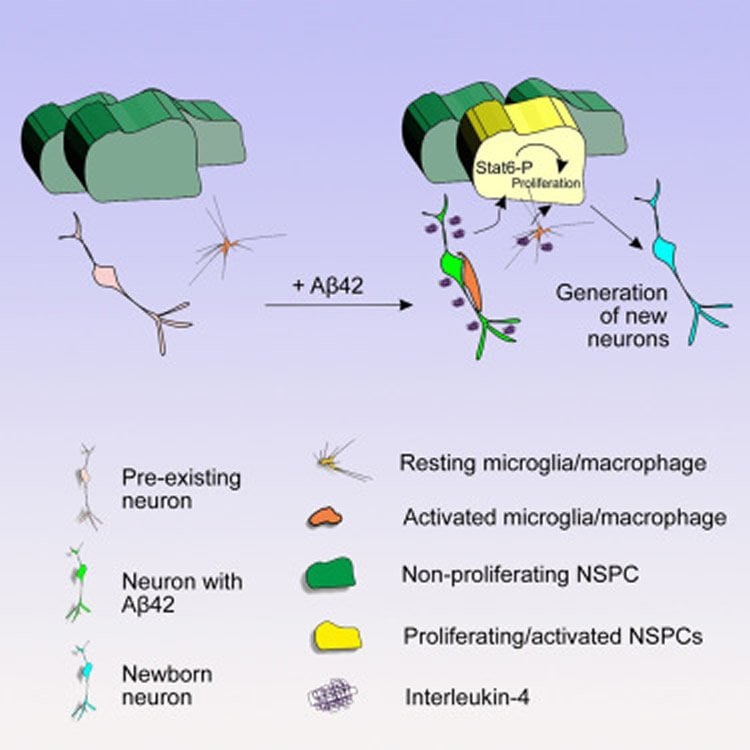
In the current study, the Dresden-based neuroscientist and his co-workers succeeded in mimicking symptoms of Alzheimer’s disease in zebrafish brain. Conditions were triggered by Amyloid-Beta42 (Aβ42) peptides – one of the major contributors to the disease in human brains. Similar to our brains, Amyloid molecules accumulated in the neurons of zebrafish leading to immune response, synaptic degeneration, cell death, learning deficits and other dysfunctions. “This is the first time, such conditions have been generated in adult zebrafish through Aβ42 deposits”, Kizil says.
The significance of neuro-immune crosstalk
The Aβ42 deposits triggered a special inflammation-related signaling pathway that ultimately lead to enhanced neurogenesis, i.e. new neurons were built. “We found that a molecule called Interleukin-4 is very much involved in the generation of neurons. The molecule is released by the dying neurons and immune system cells. It then acts on neural stem cells, which are the progenitors of neurons, by increasing their proliferation”, Kizil says. “Interleukin-4 has been known to be a player in immune response and inflammation. But to date, the direct role of IL4 on stem cell proliferation has not been shown.”
A better understanding of how we could manipulate inflammatory conditions might aid to develop novel therapies against Alzheimer’s, Kizil argues. “In humans inflammation does not seem to act as a positive cue for regeneration as it does in zebrafish. Possibly because other factors interfere in a complex manner. Our zebrafish model offers the opportunity to study such factors one by one in a reductionist manner. Besides, our study points to the significance of the immune response. That is to say: by tweaking the immune response, for example with drugs, and targeting the right cell types, we might unlock the potential of human neural stem cells to proliferate and build new neurons. Of course, the challenge remains as to what will happen to those new neurons. But first things first: we have to start from the stem cells.”
Funding: This work was supported by the Israel Science Foundation, the European Research Council, the Yeda Sela Center for Basic Research, the British Heart Foundation and the European Union, Seventh Framework Programme, Marie Curie Actions.
Source: Marcus Neitzert – DZNE
Image Source: NeuroscienceNews.com image is credited to Kizil et al./Cell Reports 2016.
Original Research: Full open access research for “IL4/STAT6 Signaling Activates Neural Stem Cell Proliferation and Neurogenesis upon Amyloid-β42 Aggregation in Adult Zebrafish Brain” by Prabesh Bhattarai, Alvin Kuriakose Thomas, Mehmet Ilyas Cosacak, Christos Papadimitriou, Violeta Mashkaryan, Cynthia Froc, Susanne Reinhardt, Thomas Kurth, Andreas Dahl, Yixin Zhang, and Caghan Kizil in Cell Reports. Published online September 22 2016 doi:10.1016/j.celrep.2016.09.075
[cbtabs][cbtab title=”MLA”]DZNE “Tweaking Immune Response May Be a Key to Combatting Alzheimer’s.” NeuroscienceNews. NeuroscienceNews, 17 October 2016.
<https://neurosciencenews.com/immune-response-neurodegeneration-5325/>.[/cbtab][cbtab title=”APA”]DZNE (2016, October 17). Tweaking Immune Response May Be a Key to Combatting Alzheimer’s. NeuroscienceNew. Retrieved October 17, 2016 from https://neurosciencenews.com/immune-response-neurodegeneration-5325/[/cbtab][cbtab title=”Chicago”]DZNE “Tweaking Immune Response May Be a Key to Combatting Alzheimer’s.” https://neurosciencenews.com/immune-response-neurodegeneration-5325/ (accessed October 17, 2016).[/cbtab][/cbtabs]
Abstract
IL4/STAT6 Signaling Activates Neural Stem Cell Proliferation and Neurogenesis upon Amyloid-β42 Aggregation in Adult Zebrafish Brain
Highlights
•Amyloid-β42 (Aβ42) forms aggregates in neurons of adult zebrafish brain
•Aβ42 causes apoptosis, inflammation, synaptic loss, and memory deficits in zebrafish
•Aβ42 leads to interleukin-4 (IL4) upregulation in neurons and microglia
•IL4/STAT6 signaling induces neural stem cell proliferation
Summary
Human brains are prone to neurodegeneration, given that endogenous neural stem/progenitor cells (NSPCs) fail to support neurogenesis. To investigate the molecular programs potentially mediating neurodegeneration-induced NSPC plasticity in regenerating organisms, we generated an Amyloid-β42 (Aβ42)-dependent neurotoxic model in adult zebrafish brain through cerebroventricular microinjection of cell-penetrating Aβ42 derivatives. Aβ42 deposits in neurons and causes phenotypes reminiscent of amyloid pathophysiology: apoptosis, microglial activation, synaptic degeneration, and learning deficits. Aβ42 also induces NSPC proliferation and enhanced neurogenesis. Interleukin-4 (IL4) is activated primarily in neurons and microglia/macrophages in response to Aβ42 and is sufficient to increase NSPC proliferation and neurogenesis via STAT6 phosphorylation through the IL4 receptor in NSPCs. Our results reveal a crosstalk between neurons and immune cells mediated by IL4/STAT6 signaling, which induces NSPC plasticity in zebrafish brains.
“IL4/STAT6 Signaling Activates Neural Stem Cell Proliferation and Neurogenesis upon Amyloid-β42 Aggregation in Adult Zebrafish Brain” by Prabesh Bhattarai, Alvin Kuriakose Thomas, Mehmet Ilyas Cosacak, Christos Papadimitriou, Violeta Mashkaryan, Cynthia Froc, Susanne Reinhardt, Thomas Kurth, Andreas Dahl, Yixin Zhang, and Caghan Kizil in Cell Reports. Published online September 22 2016 doi:10.1016/j.celrep.2016.09.075






