Summary: Synthesizing a human embryo from stem cells and using gene editing to insert the Huntingtin gene, researchers found the mutation affected the size of germ layers compared to the control embryos. Findings suggest Huntington’s disease may be a neurodevelopmental disorder that presents as a neurodegenerative disease later in life.
Source: Rockefeller University
Huntington’s disease is a fatal condition involving the death of brain cells, typically striking in midlife. But new findings suggest the disease process starts decades earlier. Although symptoms emerge in adulthood, researchers have been able to detect the earliest effects of Huntington’s in the first two weeks of human embryonic development.
The findings recast Huntington’s, often considered a neurodegenerative condition, as a developmental disease, and point to new approaches for finding treatments for a disease that currently has no cure or therapies.
“When the patient goes to the doctor, that’s when the last dominoes have fallen. But the first domino is pushed in the developmental phase,” says Ali Brivanlou, head of the Laboratory of Synthetic Embryology at Rockefeller University, who published the findings in the journal Development. “Knowing this trajectory, we may be able to block the progression of the disease.”
Early beginnings
Huntington’s is caused by mutations in a single gene, Huntingtin, resulting in the production of an unusually long protein. The gene is expressed in the fertilized egg and subsequently in every cell of the body, but its functions are not entirely known. A bigger mystery is why the defective gene appears to be detrimental only to neurons in specific parts of the brain.
Previously researchers in the Brivanlou lab found evidence that abnormalities due to Huntingtin mutation arise decades before the neurons start to perish: In the first stages of the brain development in the embryo, when uniform cells are becoming specific brain cell types and forming structures. Introducing the Huntington’s mutation in these developing cells led to abnormal neurons and structures.
Huntington’s signature
In the new study, researchers examined the effects of Huntington’s mutation at an earlier stage, called gastrulation, during which the two-week-old embryo starts to form the three embryonic germ layers, from which the progenitors of all cell types, including brain cells emerge.
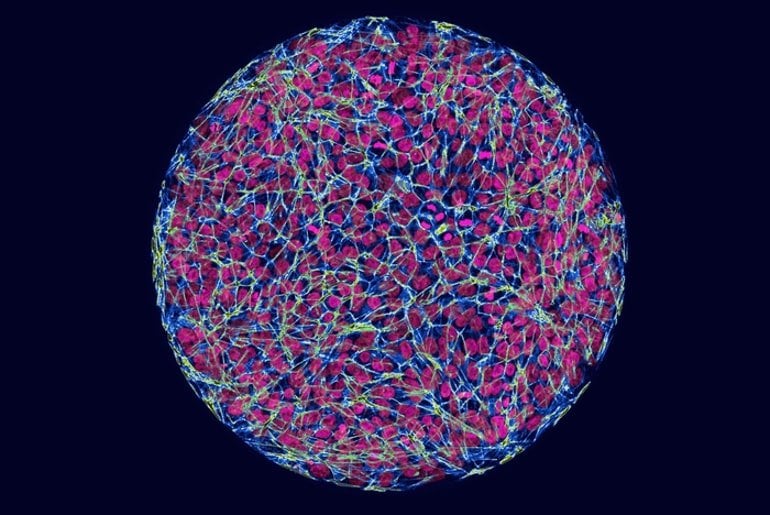
For the study, the researchers created synthetic human embryos—lab-generated embryos that are derived from stem cells and mimic the behavior of human cells during the early stages of development. They then used the gene-editing method CRISPR/Cas9 to insert the range of Huntington’s mutations found in people with the disease into the embryos.
Comparing the embryos with and without the mutation revealed a pattern: the mutations affected the size of germ layers. And the more severe mutations led to larger differences. “It’s a phenotypic signature—you can see it with your eyes,” says Brivanlou, the Robert and Harriet Heilbrunn Professor.
That visible change, the researchers found, is caused by alteration to a signaling pathway that guides the embryonic cells.
New approaches to cures
How exactly such early changes affect the development of embryos later on is unclear. But people with these mutations are born and function normally for years. Researchers suspect that a developing embryo uses certain mechanisms to compensate for the deleterious effects of Huntington’s mutations. “Understanding those mechanisms may be the key to developing new treatments that delay the symptoms, or even cure the disease,” Brivanlou says.
Using synthetic human embryos as their platform, the researchers have started to screen for drugs that can correct these abnormalities. This approach, they hope, will lead to developing clinical interventions that will address the causes of Huntington’s disease, and not just the consequences.
About this Huntington’s disease research news
Author: Katherine Fenz
Source: Rockefeller University
Contact: Katherine Fenz – Rockefeller University
Image: The image is credited to Laboratory of Stem Cell Biology and Molecular Embryology at The Rockefeller University
Original Research: Open access.
“Huntingtin CAG expansion impairs germ layer patterning in synthetic human 2D gastruloids through polarity defects” by Ali Brivanlou et al. Development
Abstract
Huntingtin CAG expansion impairs germ layer patterning in synthetic human 2D gastruloids through polarity defects
Huntington’s disease (HD) is a fatal neurodegenerative disorder caused by an expansion of the CAG repeats in the huntingtin gene (HTT). Although HD has been shown to have a developmental component, how early during human embryogenesis the HTT-CAG expansion can cause embryonic defects remains unknown.
Here, we demonstrate a specific and highly reproducible CAG length-dependent phenotypic signature in a synthetic model for human gastrulation derived from human embryonic stem cells (hESCs). Specifically, we observed a reduction in the extension of the ectodermal compartment that is associated with enhanced activin signaling.
Surprisingly, rather than a cell-autonomous effect, tracking the dynamics of TGFβ signaling demonstrated that HTT-CAG expansion perturbs the spatial restriction of activin response. This is due to defects in the apicobasal polarization in the context of the polarized epithelium of the 2D gastruloid, leading to ectopic subcellular localization of TGFβ receptors.
This work refines the earliest developmental window for the prodromal phase of HD to the first 2 weeks of human development, as modeled by our 2D gastruloids.






