Summary: A lipid nanocarrier small enough to pass the blood-brain barrier could be an effective method for delivering chemotherapy medications to help fight glioblastoma brain tumors, a new study reports.
Source: Medical University of South Carolina.
Nanocarrier provides efficient delivery of chemotherapeutic drug.
Great discoveries do come in small packages. Few know that better than Ann-Marie Broome, Ph.D., who feels nanotechnology holds the future of medicine with its ability to deliver powerful drugs in tiny, designer packages.
Her latest research finds the perfect application – targeting cancerous brain tumor cells.
Results from her recent paper published online in the international journal Nanomedicine – Future Medicine found that a lipid nanocarrier engineered to be small enough to get past the blood-brain barrier could be targeted to deliver a chemotherapeutic drug more efficiently to tumor cells in the brain. In vivo studies showed specific uptake and increased killing in glial cells, so much so that Broome initially questioned the results.
“I was very surprised by how efficiently and well it worked once we got the nanocarrier to those cells,” she said, explaining that initial results were so promising that she had her team keep repeating the experiments, using different cell lines, dosage amounts and treatment times.Researchers and clinicians are excited because it potentially points the way to a new treatment option for patients with certain conditions, such as glioblastoma multiforme (GBM), the focus of this study.
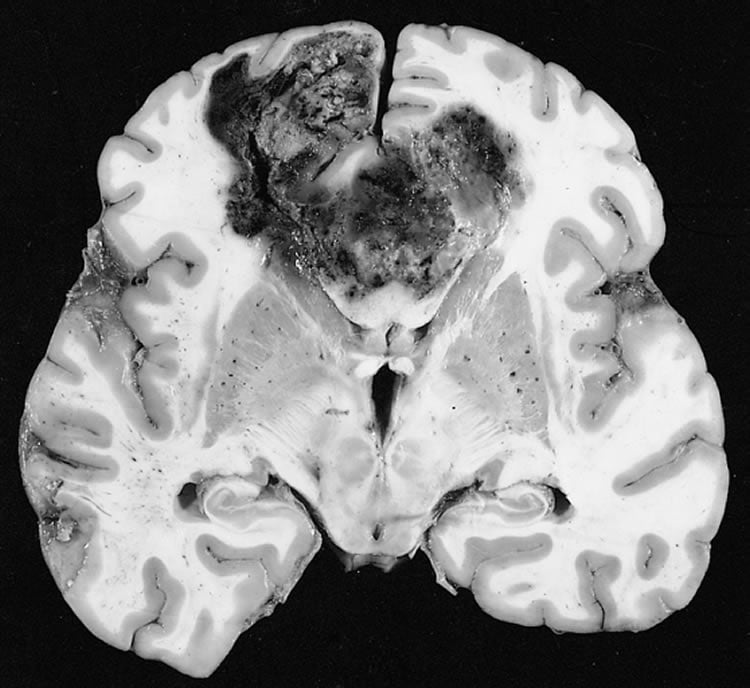
Glioblastoma multiforme is a devastating disease with no curative options due to several challenges, said Broome, who is the director of Molecular Imaging of the Medical University of South Carolina’s Center for Biomedical Imaging and director of Small Animal Imaging of Hollings Cancer Center. The brain tumor has a significant overall mortality, in part due to its location, difficulty of surgical treatment and the inability to get drugs through the blood-brain barrier, a protective barrier designed to keep a stable environment within and surrounding the brain.
In 40 percent of cases, standard treatments will extend life expectancy 4 to 7 months. “It’s really a dismal outcome. There are better ways to deliver standard of care.”
That’s where Broome and her nanotechnology lab enter in.
Nanotechnology is medicine, engineering, chemistry, and biology all bundled together and conducted at the nanoscale, between the range of 1 to 1,000 nanometers. For comparison, a thin newspaper page is about 100,000 nanometers thick. Broome and her team took what they know about the cancer’s biology and of platelet-derived growth factor (PDGF), one of numerous growth factor proteins that regulates cell growth and division and is also overexpressed on tumor cells in the brain. With that in mind, they engineered a micelle that is a phospholipid nanocarrier, “a bit of fat globule,” to deliver a concentrated dose of the chemotherapy drug temozolomide (TMZ) to the GBM tumor cells.
“Micelles of a certain size will cross the blood-brain barrier carrying a concentrated amount of TMZ,” she explained about how the nanotechnology works. “The PDGF is used much like a postal address. The micelle gets it to the street, and the PDGF gets it to the house.” This targeting ability is important because researchers have learned that it’s likely that the GBM will recur, she said.
“It’s thought that satellite cells left behind after surgical removal are the fastest growing and most dangerous ones. We’re trying to kill those rapidly growing satellite cells that will grow into new tumors in that location or others. These satellite tumors grow more aggressively than others. You have to hit them hard, fast and aggressively.”
Surprisingly, nanotechnology is already a part of everyday life in many ways that people don’t realize. It’s used in everything from makeup as moisturizers or UV sunscreens to ice cream to maintain frozen temperatures and creamy textures.
In medicine, Broome said, researchers construct nanocarriers that are stable and stealthy. “Your immune cells can’t attack them. They remain hidden.”When the package gets to where it’s going, nanotechnologists have various methods to get the micelles to release their payloads- one way is to use the acidic nature of a rapidly growing tumor. In normal circulation, the pH of blood is slightly alkaline and the micelle stays intact. What researchers have discovered is that in many tumor types, the pH drastically changes to an acidic environment.
“While the tumor is growing, it creates waste by-products and metabolites that alter the pH, thus lowering it. As the center becomes more necrotic, it becomes even more acidic.”
The change in pH triggers a release of the drug from our micelles just where clinicians want it to go to reduce toxicity to the rest of the body, she said.
“We take advantage of the tumor’s natural environment as well as the cellular expression. I’m a big proponent of understanding that microenvironment has an impact on how well you can treat tumors. It’s probably why so many therapeutics fail – because you have to take into account the immune system, the local environment, and the cells themselves – all three of these are important considerations.”
That’s why nanotechnology has an edge in shaping future cancer treatments.
“It’s very important that the public recognize that nanotechnology is the future. It impacts so many different fields. It has a clear impact on cancer biology and potentially has an impact on cancers that are inaccessible, untreatable, undruggable – that in normal circumstances are ultimately a death knell.”All too familiar with this is researcher and clinician Amy Lee Bredlau, M.D., director of MUSC Health’s Pediatric Brain Tumor Program, who also was a part of the study. Broome said she relishes having a clinician’s perspective in the lab to focus the group on translational outcomes for the patients.
“That’s why it’s so gratifying working with Amy Lee. She works with many cancers for which there are no options. We’re trying to provide options.”
Bredlau agreed. “This paper is exciting because it demonstrates a novel approach to treating brain tumors, combining nanotechnology targeting to a marker of brain tumors with a specialized delivery system. It will allow us eventually to target aggressive childhood and adult brain tumors.”
Bredlau said she’s taking time out from her clinical practice to be in Broome’s research lab because she knows that’s how she can best accelerate the process.
“I am passionate about improving the lives of my patients, now and in the future. Advancing research now is the best way to improve the lives of my patients to come.”
Bredlau sees nanotechnology as having the power to revolutionize treatment for brain tumors. “When we perfect this strategy, we will be able to deliver potent chemotherapies only to the area that needs them. This will dramatically improve our cure rates while cutting out a huge portion of our side effects from chemotherapy. Imagine a world where a cancer diagnosis not only was not life-threatening, but also did not mean that you would be tired, nauseated or lose your hair.”
Though excited by the study’s results, Broome cautions that there’s much more work to be done before new treatment options are readily available for patients.
“It may or may not be effective for all types of GBMs. There are subtypes as well as therapeutic-resistant GBMs that these nanocarriers may not impact. We need to continue rigorous testing to verify and validate our initial findings.”
They will be exploring an expanding field of targeted biomarkers available for GBM tumor cells. As is common in breast cancer and other cancer types, this cancer has specific cell surface receptors that are overexpressed, she said.
And though the drug TMZ in this protocol works very efficiently, it may not be the best drug for the majority of the people, she said. “Now that we know we can get the drug to its designated location and get it to work efficiently, we have a comparator. We can test more lethal and different combinations of drugs that have never before been used in this scenario.”
This method of drug delivery also opens new windows to immunotherapy treatments garnering recognition internationally. Broome wants to take chemotherapeutics and combine them with new immuno-therapeutic treatments to form unique combination delivery packages.
It’s ambitious.
Broome, whose team jokes that she keeps “a long, running list of impossible tasks,” said the work also translates to so many fields beyond cancer including stroke, transplant and regenerative medicine, where it could be used for example in wound healing in dermatology or organ maintenance in transplantation. It’s one reason she submitted her latest research to an international journal because she wants to accelerate advances in nanotechnology, a field she has no doubt will change how medicine is done.
“They are the primary reason I continue to do what I do,” she said of the patients who face grim diagnoses. “They give me hope. The possibilities for nanotherapeutics are endless and bright.”
Funding: Funding information not available.
Source: Dawn Brazell – Medical University of South Carolina
Image Source: This NeuroscienceNews.com image is in the public domain.
Original Research: Abstract for “Delivery of a drug cache to glioma cells overexpressing platelet-derived growth factor receptor using lipid nanocarriers” by Kayla Miller, Suraj Dixit, Amy-Lee Bredlau, Alfred Moore, Emilie McKinnon, and Ann-Marie Broome in Nanomedicine – Future Medicine. Published online March 2016 doi:10.2217/nnm.15.218
[cbtabs][cbtab title=”MLA”]Medical University of South Carolina. “Tiny Packages May Pack Powerful Treatment for Brain Tumors.” NeuroscienceNews. NeuroscienceNews, 22 May 2016.
<https://neurosciencenews.com/glioblastoma-tmz-bbb-4268/>.[/cbtab][cbtab title=”APA”]Medical University of South Carolina. (2016, May 22). Tiny Packages May Pack Powerful Treatment for Brain Tumors. NeuroscienceNews. Retrieved May 22, 2016 from https://neurosciencenews.com/glioblastoma-tmz-bbb-4268/[/cbtab][cbtab title=”Chicago”]Medical University of South Carolina. “Tiny Packages May Pack Powerful Treatment for Brain Tumors.” NeuroscienceNews.
https://neurosciencenews.com/glioblastoma-tmz-bbb-4268/ (accessed May 22, 2016).[/cbtab][/cbtabs]
Abstract
Asymmetric division of clonal muscle stem cells coordinates muscle regeneration in vivo
Aim: Glioblastoma multiforme is a devastating disease with no curative options due to the difficulty in achieving sufficient quantities of effective chemotherapies into the tumor past the blood–brain barrier. Micelles loaded with temozolomide (TMZ) were designed to increase the delivery of this drug into the brain.
Materials & methods: pH-responsive micelles composed of distearoyl phosphoethanolamine-PEG-2000-amine and N-palmitoyl homocysteine were surface-functionalized with PDGF peptide and Dylight 680 fluorophore.
Results & conclusion: PDGF-micelles containing TMZ have specific uptake and increased killing in glial cells compared with untargeted micelles. In vivo studies demonstrated selective accumulation of PDGF-micelles containing TMZ in orthotopic gliomas implanted in mice. Targeted micelle-based drug carrier systems hold potential for delivery of a wide variety of hydrophobic drugs thereby reducing its systemic toxicity.
“Delivery of a drug cache to glioma cells overexpressing platelet-derived growth factor receptor using lipid nanocarriers” by Kayla Miller, Suraj Dixit, Amy-Lee Bredlau, Alfred Moore, Emilie McKinnon, and Ann-Marie Broome in Nanomedicine – Future Medicine. Published online March 2016 doi:10.2217/nnm.15.218






