Summary: In the emerging field of cancer neuroscience, researchers have typically viewed the nervous system as a “gas pedal” that accelerates tumor progression. However, a new study has identified a surprising “brake.”
Investigating melanoma in mouse models, the team discovered that sympathetic nerves, the same ones responsible for the “fight-or-flight” response, can actually inhibit tumor growth. By releasing norepinephrine, these nerves flip a molecular switch on immune cells, preventing the tumor from recruiting the cellular allies it needs to thrive.
Key Facts
- The Sympathetic Brake: While sensory (pain) nerves were found to promote melanoma growth, sympathetic nerves had the opposite effect, actively slowing the expansion of the tumor.
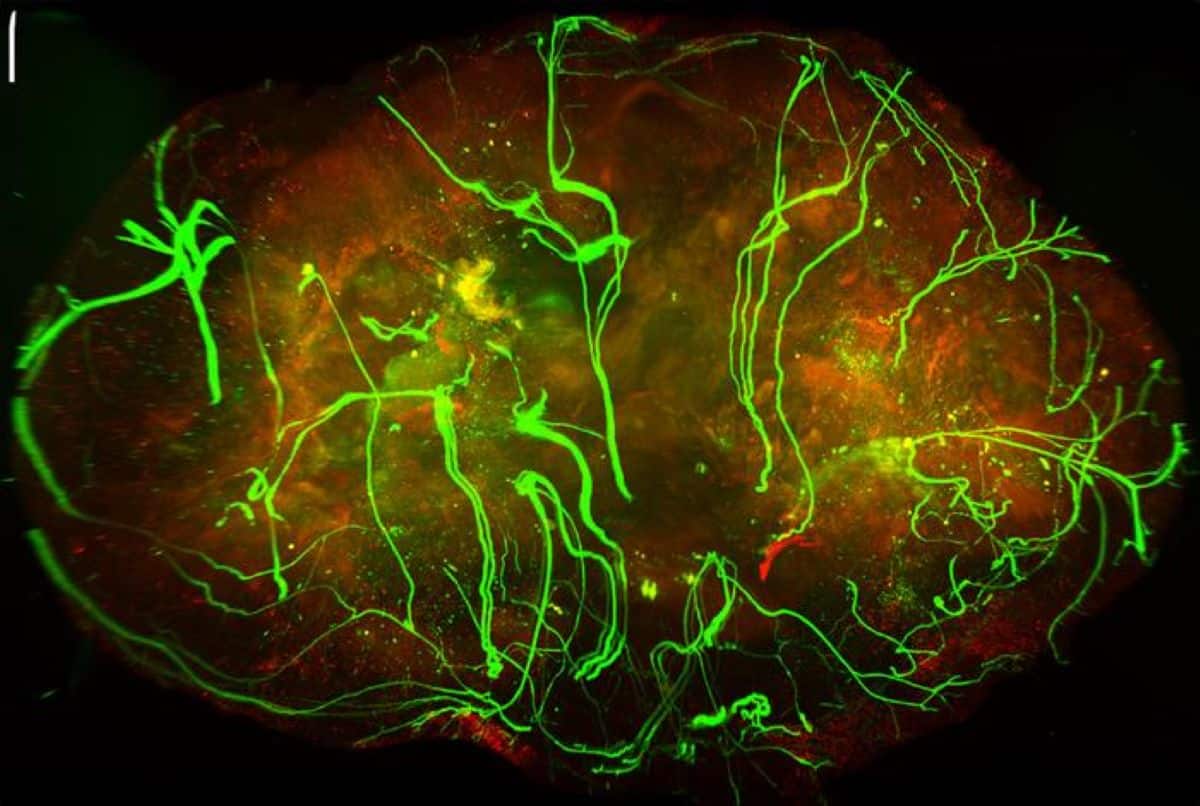
- Whole-Mount Immuno-Labeling: Researchers used a cutting-edge technique to make entire tissue samples optically transparent. This allowed them to 3D-trace every nerve fiber as it wove its way into the melanoma.
- Targeting Macrophages: The anti-tumor effect works through alpha-adrenergic receptors on macrophages (a type of immune cell). Normally, tumors “reprogram” these macrophages to support cancer growth; however, sympathetic signaling reduces the number of these pro-tumor allies.
- Norepinephrine’s Dual Role: In the skin, sympathetic nerves release norepinephrine. While this hormone is often associated with stress, in the context of melanoma, it signals the immune system to stay in an anti-tumor mode.
- Drug Repurposing Potential: Medications that target alpha-adrenergic receptors are already FDA-approved and widely used to treat high blood pressure, raising the possibility of repurposing these drugs for cancer therapy.
Source: Weill Cornell University
Nerve fibers within melanomas can slow the growth of these tumors, according to a study led by Weill Cornell Medicine investigators.
The findings help clarify the emerging field of cancer neuroscience and may inform future therapeutic strategies.
In the study, published April 29 in Neuron, the researchers used mouse models of the skin cancer melanoma to examine the presence and the effects of peripheral nerves that grow into tumors.
They found that nerves of the sympathetic nervous system are often abundant in such tumors, and can inhibit tumor growth by reducing local tumor-supportive macrophages—a type of immune cell.
“The nervous system typically has been considered as a driver of cancer growth, but here we’ve found that it can be a brake on cancer growth in some contexts,” said study senior author Dr. David J. Simon, an assistant professor of biochemistry and biophysics at Weill Cornell Medicine. “Now the key will be to see how broadly relevant this is for human cancers, and how we can best step on that brake to help cancer patients.”
The peripheral nervous system is the tree-like system of nerves that extends outside the brain and spinal cord. It includes the sensory nerves that underlie feelings such as heat and cold, pain and itch; and nerves of the sympathetic nervous system, which transmit signals from the brain to influence the functions of various organs.
In the skin, most sympathetic nerve fibers can release the stress hormone norepinephrine, affecting immune cells, sweat glands and other targets as part of the “fight-or-flight” response.
Peripheral nerves are commonly found in tumors, but it is only in recent years that researchers have begun examining the roles of these nerves in cancer outcomes. Most of these investigations have found that sensory and sympathetic nerves can enhance tumor growth, for example by releasing molecules that suppress antitumor immunity. In just the last few years, however, there have been hints that peripheral nerves in some cases may be able to slow tumor growth instead.
Dr. Simon and his team have expertise in studying the growth and survival of peripheral nerve fibers, especially those that grow into the skin.
“We knew that these nerves entered melanoma, but studying their role in cancer growth was not our main goal,” said Dr. Simon. “But we were fortunate to receive generous early-stage support from the Pershing Square Sohn Cancer Research Alliance that allowed us to take a risk and explore these nerve-tumor interactions in detail.”
“We used a technique called whole mount immuno-labeling, in which an entire tissue sample is made optically transparent, to count, identify and trace the paths of the nerves in the tumors,” said study first author Dr. Tingting Liu, a postdoctoral research associate in the Simon Lab.
These initial investigations revealed that pain-sensitive nerves and sympathetic nerves were prevalent in the melanomas, increasing in number as the tumors grew, particularly in slower-growing tumors.
The pain-sensitive nerves, consistent with prior studies, appeared to have a pro-tumor effect—depleting them inhibited tumor growth—but the sympathetic nerves surprisingly exerted an anti-tumor effect.
The sympathetic nerves identified in the study release norepinephrine, which can activate receptors called adrenergic receptors on other cells in the vicinity. The researchers traced the anti-tumor effect in their models to a subset of adrenergic receptors called alpha adrenergic receptors, and identified immune cells called macrophages as the key cellular targets of this alpha adrenergic signaling.
Tumors often turn macrophages into allies, for example by switching them into an immunosuppressive mode, but the alpha adrenergic signaling reduced the numbers of such pro-tumor macrophages, thereby slowing tumor growth.
The findings open up the prospect of future anti-cancer therapies that target sympathetic nerves within tumors, or even the alpha adrenergic receptors on tumor-associated macrophages.
Drugs targeting these receptors are already in use as common blood pressure medicines. For now, Dr. Simon plans to continue with more fundamental research, for example to tease apart how these adrenergic receptors are activated and signal in actual cancers in humans.
“There’s a lot still to do in terms of the basic biology here,” he said.
Funding: The research reported in this story was supported in part by the National Cancer Institute, part of the National Institutes of Health, through grant numbers R01CA256188, R01CA272717, P30CA08748, P30CA014520 and R37CA300434, and by the U.S. Department of Defense through grant number ME240045. Additional support was provided by the Fernholz Family Foundation and the Pershing Square Sohn Cancer Research Alliance.
Key Questions Answered:
A: It’s more complicated than that. While the sympathetic nervous system is part of the stress response, “chronic stress” usually involves systemic hormones that can suppress the immune system. This study focuses on the local release of norepinephrine by nerve fibers directly inside the tumor. It’s about the “hard-wiring” of the nerves, not the psychological feeling of stress.
A: It comes down to the chemical signals they send. Pain nerves release molecules that can blind the immune system to the tumor. Sympathetic nerves release norepinephrine, which specifically blocks the tumor’s ability to “recruit” macrophages to its side.
A: Since alpha-adrenergic drugs are already in use for blood pressure, clinical trials could move faster than they would for a brand-new molecule. However, we first need to confirm if this “brake” exists in human melanomas the same way it does in mouse models.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this neuroscience and cancer research news
Author: Corinne Esposito
Source: Weill Cornell Medicine
Contact: Corinne Esposito – Weill Cornell Medicine
Image: The image is credited to Dr. David J. Simon
Original Research: Open access.
“A local sympathetic-immune axis inhibits melanoma growth in mice by dictating adrenergic control” by Tingting Liu, Daniel Y. Kutsovsky, Ethan M. Earlie, Liangliang Ji, Michael Iskols, Shakti Ramsamooj, Xavier I. Dawkins, Marwa Zerhouni, Alexander Birbrair, Elena Piskounova, Ming O. Li, Ashley M. Laughney, and David J. Simon. Neuron
DOI:10.1016/j.neuron.2026.04.016
Abstract
A local sympathetic-immune axis inhibits melanoma growth in mice by dictating adrenergic control
The nervous system drives tumor growth directly through intra-tumoral axons and indirectly through the systemic action of hormones. Yet contexts where the nervous system inhibits tumor growth are less defined.
Here, we performed optical reconstruction of axonal innervation in mouse models of cutaneous melanoma, revealing progressive innervation by sympathetic axons. Local depletion of these axons accelerates while local optogenetic activation slows melanoma growth, together consistent with these axons acting as a physiological growth brake.
The sympathetic nervous system is typically associated with driving tumor growth through activation of β-adrenergic receptors (ARs).
Here, we find that the initial tumor seeding conditions sensitize melanomas from βAR-driven growth promotion toward α2-AR-driven growth inhibition. Mechanistically, the axonal activation of α2 ARs restricts the number and distribution of pro-tumor myeloid cells, independently of T cell activity.
Together, our data reveal context-dependent, bidirectional neural control of tumor progression.