Summary: A metabolite produced by lactic acid bacteria binds to the third hydroxycarboxylic acid receptor (HCA), signalling the immune system. Researchers believe the receptor evolved to allow great apes to consume foods that were starting to decay. They suggest the receptor could mediate some beneficial and anti-inflammatory effects of lactic acid in humans, and could serve as a target to treat inflammatory diseases.
Source: PLOS
Researchers have discovered that humans and great apes possess a receptor on their cells that detects metabolites from bacteria commonly found in fermented foods and triggers movement of immune cells. Claudia Stäubert of the University of Leipzig and colleagues report these findings in a new study published 23rd May in PLOS Genetics.
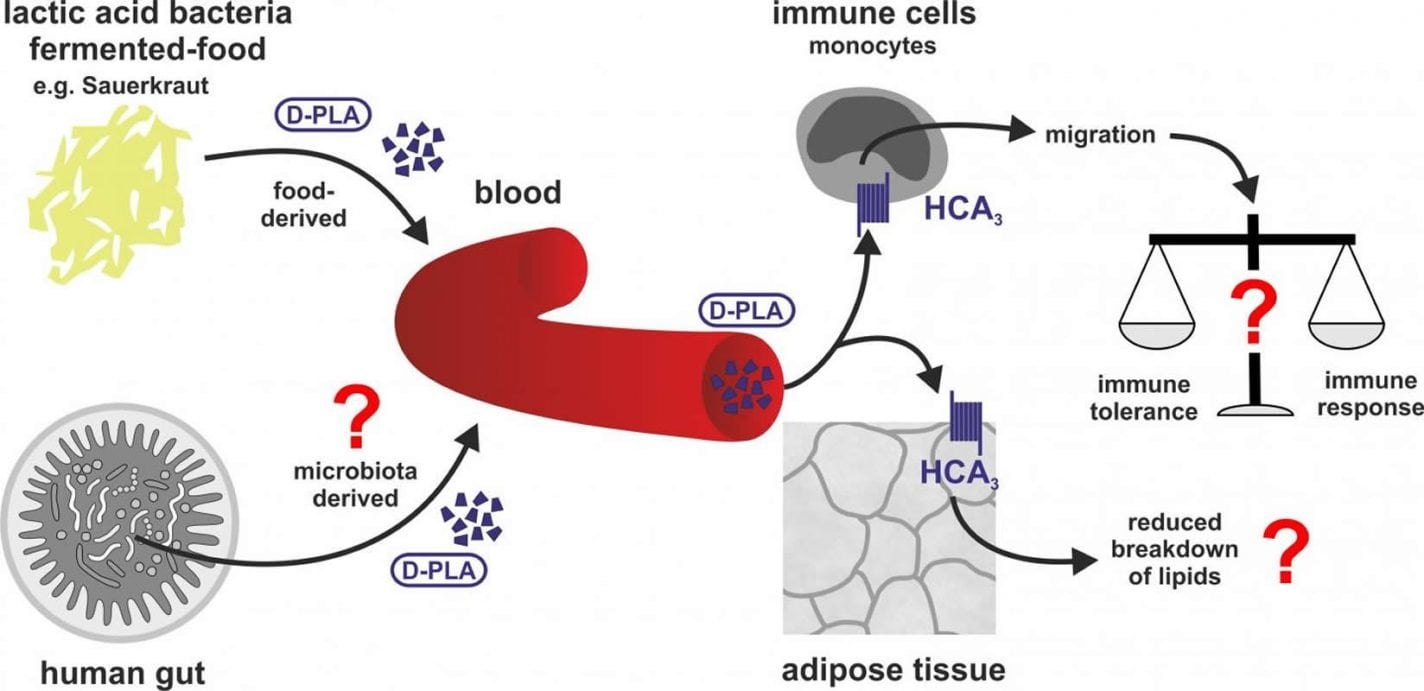
Consuming lactic acid bacteria – the kind that turn milk into yogurt and cabbage into sauerkraut – can offer many health benefits, but scientists still don’t understand, on a molecular level, why it is helpful to ingest these bacteria and how that affects our immune system. Now, Stäubert and her colleagues have found one way that lactic acid bacteria interact with our bodies. Initially the researchers were investigating proteins on the surface of cells called hydroxycarboxylic acid (HCA) receptors. Most animals have only two types of this receptor but humans and great apes have three. The researchers discovered that a metabolite produced by lactic acid bacteria, D-phenyllactic acid, binds strongly to the third HCA receptor, signalling the immune system their presence. The researchers propose that the third HCA receptor arose in a common ancestor of humans and great apes, and enabled them to consume foods that are starting to decay, such as fruits picked up from the ground.
The study yields new insights into the evolutionary dynamics between microbes and their human hosts and opens new research directions for understanding the multiple positive effects of eating fermented foods. “We are convinced that this receptor very likely mediates some beneficial and anti-inflammatory effects of lactic acid bacteria in humans,” stated author Claudia Stäubert. “That is why we believe it could serve as a potential drug target to treat inflammatory diseases.”
Future studies may reveal the details of how D-phenyllactic acid impacts the immune system, and whether the metabolite also affects fat cells, which also carry the third HCA receptor on their surfaces.
Source:
PLOS
Media Contacts:
Claudia Stäubert – PLOS
Image Source:
The image is credited to Claudia Stäubert.
Original Research: Open access
“Metabolites of lactic acid bacteria present in fermented foods are highly potent agonists of human hydroxycarboxylic acid receptor 3”. Anna Peters, Petra Krumbholz, Elisabeth Jäger, Anna Heintz-Buschart, Mehmet Volkan Çakir, Sven Rothemund, Alexander Gaudl, Uta Ceglarek, Torsten Schöneberg, Claudia Stäubert.
PLOS Genetics. doi:10.1371/journal.pgen.1008145
Abstract
Metabolites of lactic acid bacteria present in fermented foods are highly potent agonists of human hydroxycarboxylic acid receptor 3
The interplay of microbiota and the human host is physiologically crucial in health and diseases. The beneficial effects of lactic acid bacteria (LAB), permanently colonizing the human intestine or transiently obtained from food, have been extensively reported. However, the molecular understanding of how LAB modulate human physiology is still limited. G protein-coupled receptors for hydroxycarboxylic acids (HCAR) are regulators of immune functions and energy homeostasis under changing metabolic and dietary conditions. Most mammals have two HCAR (HCA1, HCA2) but humans and other hominids contain a third member (HCA3) in their genomes. A plausible hypothesis why HCA3 function was advantageous in hominid evolution was lacking. Here, we used a combination of evolutionary, analytical and functional methods to unravel the role of HCA3 in vitro and in vivo. The functional studies included different pharmacological assays, analyses of human monocytes and pharmacokinetic measurements in human. We report the discovery of the interaction of D-phenyllactic acid (D-PLA) and the human host through highly potent activation of HCA3. D-PLA is an anti-bacterial metabolite found in high concentrations in LAB-fermented food such as Sauerkraut. We demonstrate that D-PLA from such alimentary sources is well absorbed from the human gut leading to high plasma and urine levels and triggers pertussis toxin-sensitive migration of primary human monocytes in an HCA3-dependent manner. We provide evolutionary, analytical and functional evidence supporting the hypothesis that HCA3 was consolidated in hominids as a new signaling system for LAB-derived metabolites.






