Summary: Metformin, a common diabetes medication, shows promise for the treatment of anxiety and depression.
Source: SfN
The antidiabetic medication metformin reduces anxiety-like behaviors in male mice by increasing serotonin availability in the brain, according to a study published in Journal of Neuroscience. These findings could have implications for the treatment of patients with both metabolic and mental disorders.
People with diabetes have an increased risk for mood disorders such as depression. Although the mechanisms underlying the relationship between insulin resistance — the precursor to diabetes — and depression are not known, studies suggest the neurotransmitter serotonin may be the culprit.
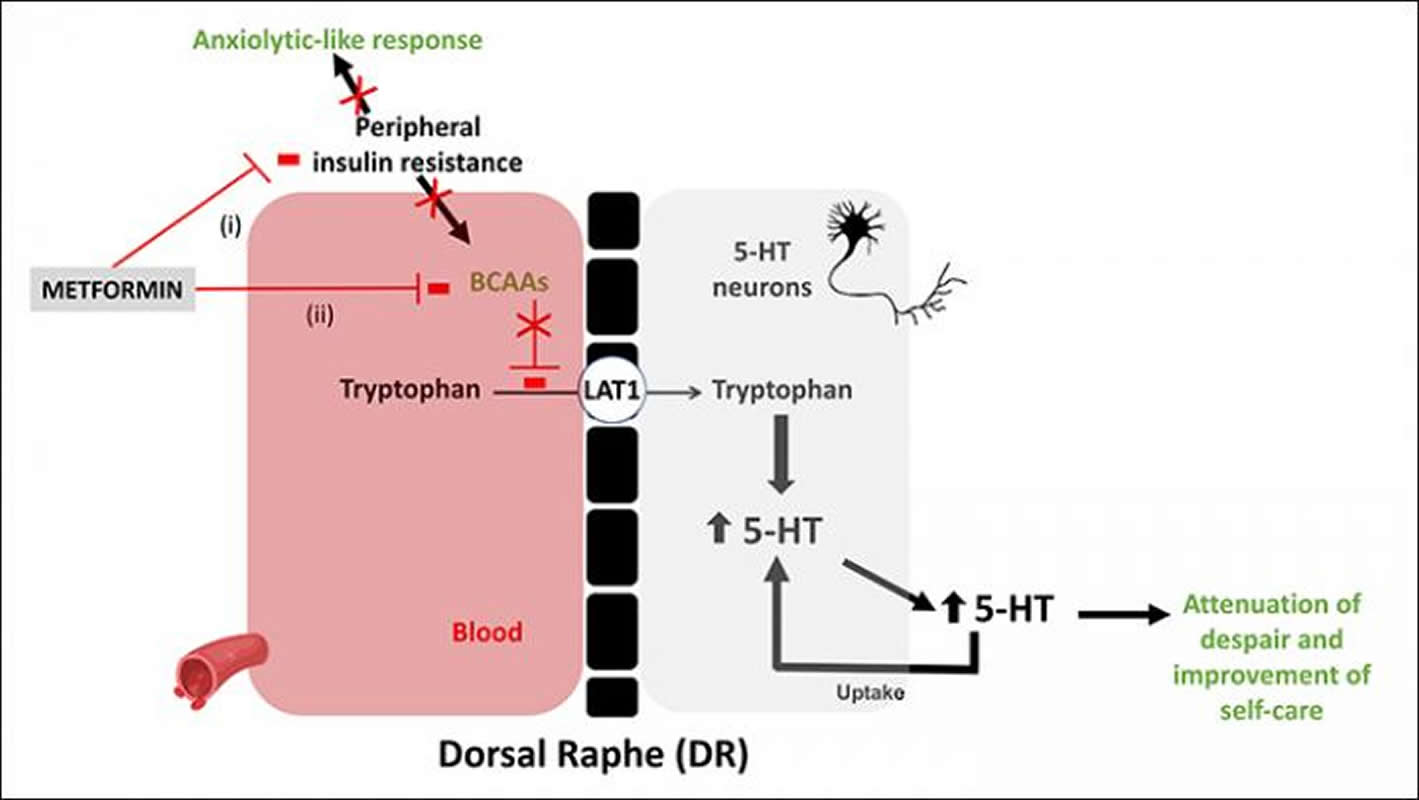
In mice raised on a high-fat diet, Bruno Guiard and colleagues demonstrate that the insulin-sensitizing drug metformin reduces levels of amino acids that impair the entry of tryptophan in the brain and thereby limit its conversion into serotonin. The drug’s antidepressant-like effects were accompanied by improved neurotransmission in the hippocampus. The researchers achieved similar effects by reducing the amount of so-called branched chained amino acids in the diet.
Source:
SfN
Media Contacts:
David Barnstone – SfN
Image Source:
The image is credited to Zemdegs et al., JNeurosci (2019).
Original Research: Closed access
“Metformin promotes anxiolytic and antidepressant-like responses in insulin-resistant mice by decreasing circulating branched-chain amino acids”. J Zemdegs, H Martin, H Pintana, S Bullich, S Manta, MA Marqués, C Moro, S Layé, F Ducrocq, N Chattipakorn, SC Chattipakorn, C Rampon, L Pénicaud, X Fioramonti and BP Guiard.
Journal of Neuroscience. doi:10.1523/JNEUROSCI.2904-18.2019
Abstract
Metformin promotes anxiolytic and antidepressant-like responses in insulin-resistant mice by decreasing circulating branched-chain amino acids
Epidemiological studies indicate that insulin resistance (IR), a hallmark of Type 2 diabetes, is associated with an increased risk of major depression. Here, we demonstrated that male mice fed a high fat diet (HFD) exhibited peripheral metabolic impairments reminiscent of IR accompanied by elevated circulating levels of branched-chain amino acids (BCAAs) whereas both parameters were normalized by a chronic treatment with metformin (Met). Given the role of BCAAs in the regulation of tryptophan influx into the brain, we then explored the activity of the serotonin (5-HT) system. Our results indicated that HFD mice displayed impairment in the electrical activity of dorsal raphe (DR) 5-HT neurons, attenuated hippocampal extracellular 5-HT concentrations and caused anxiety, one of the most visible and early symptoms of depression. On the contrary, Met stimulated 5-HT neurons excitability and 5-HT neurotransmission while hindering HFD-induced anxiety. Met also promoted antidepressant-like activities as observed with fluoxetine. In light of these data, we designed a modified HFD in which BCAA dietary supply was reduced by half. Deficiency in BCAAs failed to reverse HFD-induced metabolic impairments while producing antidepressant-like activity and enhancing the behavioral response to fluoxetine. Our results suggest that Met may act by decreasing circulating BCAAs levels to favor serotonergic neurotransmission in the hippocampus and promote antidepressant-like effects in mice fed a HFD. These findings also lead us to envision that a diet poor in BCAAs, provided either alone or as add-on therapy to conventional antidepressant drugs, could help relieving depressive symptoms in patients with metabolic comorbidities.
Significance statement:
Insulin resistance in humans is associated with increased risk of anxio-depressive disorders. Such relationship has been also found in rodents fed a High Fat Diet (HFD). To determine whether insulin-sensitizing strategies induce anxiolytic- and/or antidepressant-like activities and to investigate the underlying mechanisms, we tested the effects of metformin, an oral antidiabetic drug, in mice fed a HFD. Metformin reduced levels of circulating branched-chain amino acids which regulate tryptophan uptake within the brain. Moreover, metformin increased hippocampal serotonergic neurotransmission while promoting anxiolytic- and antidepressant-like effects. Moreover, a diet poor in these amino acids produced similar beneficial behavioral property. Collectively, these results suggest that metformin could be used as add-on therapy to conventional antidepressant for the comorbidity between metabolic and mental disorders.