Summary: FIASMA antidepressants, such as amitriptyline and desipramine, halt the growth of four different kinds of bacterial pathogens in cell cultures and animal models. The antidepressants have shown to be effective in killing intracellular bacteria in two chlamydia infections, as well as human granulocytic anaplasmosis, a tick-borne disease that attacks white blood cells.
Source: Virginia Commonwealth University
Some antidepressants could potentially be used to treat a wide range of diseases caused by bacteria living within cells, according to work by researchers in the Virginia Commonwealth University School of Medicine and collaborators at other institutions.
Research published in the April print edition of the journal Life Science Alliance shows that antidepressant drugs called FIASMAs, including desipramine, amitriptyline, and nortriptyline, halt the growth or kill four different intracellular bacterial pathogens in tissue cell culture and animal models.
“Antibiotic options for diseases caused by intracellular bacteria are limited because many of these drugs cannot penetrate our cell membranes. In essence, the bacteria are protected,” said Jason Carlyon, Ph.D., leader of the study and professor in the VCU Department of Microbiology and Immunology.
Tetracycline antibiotics are most commonly prescribed to treat intracellular bacterial infections because they can cross cell membranes to reach the microbes. However, tetracyclines can cause allergic reactions in some patients and physicians advise against their use by pregnant women and children due to undesirable side effects. Additionally, antibiotic resistance in some intracellular bacteria has been reported.
“It would be highly beneficial to have a class of drugs to treat such diseases in patients for whom tetracyclines are contraindicated,” Carlyon said. “These drugs could provide an alternative to antibiotics or even be used in conjunction with them as an augmentation approach to treat infections that typically require prolonged courses of antibiotic therapy, such as those caused by Chlamydia pneumoniae and Coxiella burnetti.”
The team of researchers from VCU, Indiana University Medical Center, University of Nebraska Medical Center, University of Arkansas for Medical Sciences, and the University of South Florida, including Carlyon and lead author Chelsea Cockburn, an M.D.-Ph.D. candidate, are the first to investigate the mechanisms by which FIASMAs target multiple intracellular bacteria in detail.
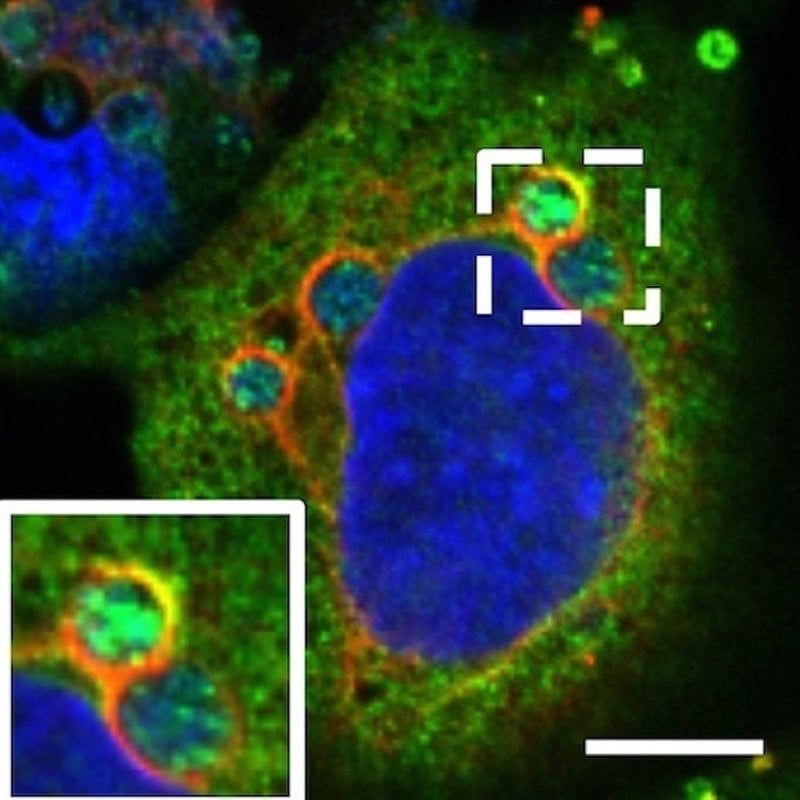
The scientists tested FIASMA susceptibility for four bacterial species that cause human granulocytic anaplasmosis, a tick-borne disease that attacks white blood cells called neutrophils and can be fatal to immune compromised individuals; Q fever, a debilitating pneumonic disease; and two chlamydia infections.
FIASMAs ultimately disrupt how cholesterol, a key nutrient utilized by many intracellular pathogens, traffics inside cells to alter bacterial access to the lipid. The researchers first proved FIASMA treatment efficacy by halting anaplasmosis in both tissue culture and mice. Next, they extended their observations to demonstrate that FIASMA treatment killed the Q fever agent,Coxiella burnetii, and partially inhibited chlamydial infections in cell culture.
“Since FIASMAs influence cholesterol trafficking in the cell and cholesterol plays a role in so many facets of our biology, they have been used to treat a wide variety of conditions and diseases,” Carlyon said.
He added that the effect of FIASMAs on intracellular cholesterol ultimately bypasses the need to directly target the bacteria.
“What is so exciting about this study is that the class of drugs we evaluated targets an enzyme in our cells regulating cholesterol, not the bacteria,” Carlyon said. “I do not envision the pathogens being able to develop resistance to this treatment because it is targeting a host pathway that they very much need to grow and survive inside of the body.”
Other investigators involved in the research included Rebecca Martin, Ph.D., and Daniel Conrad, Ph.D., (VCU), Charles Chalfant, Ph.D., (University of South Florida), Daniel Voth, Ph.D., (University of Arkansas for Medical Sciences), Elizabeth Rucks, Ph.D., (University of Nebraska Medical Center) and Stacey Gilk, Ph.D., (Indiana University School of Medicine).
Funding: Research was supported by grants from the NIH-National Institute of Allergy and Infectious Diseases, including 1R01 AI139072, 2R01 AI072683, 5R01 AI018697, 1R01 AI139176, 1R21 AI127931, and 1R21 AI121786, and from the NIH-National Institute of General Medicine Sciences (1P20 GM103625).
Source:
Virginia Commonwealth University
Media Contacts:
Tom Gresham – Virginia Commonwealth University
Image Source:
The image is credited to Life Science Alliance.
Original Research: Open access
“Functional inhibition of acid sphingomyelinase disrupts infection by intracellular bacterial pathogens”. Chelsea L Cockburn, Ryan S Green, Sheela R Damle, Rebecca K Martin, Naomi N Ghahrai, Punsiri M Colonne, Marissa S Fullerton, Daniel H Conrad, Charles E Chalfant, Daniel E Voth, Elizabeth A Rucks, Stacey D Gilk, Jason A Carlyon .
Life Science Alliance. doi:10.26508/lsa.201800292
Abstract
Functional inhibition of acid sphingomyelinase disrupts infection by intracellular bacterial pathogens
Intracellular bacteria that live in host cell–derived vacuoles are significant causes of human disease. Parasitism of low-density lipoprotein (LDL) cholesterol is essential for many vacuole-adapted bacteria. Acid sphingomyelinase (ASM) influences LDL cholesterol egress from the lysosome. Using functional inhibitors of ASM (FIASMAs), we show that ASM activity is key for infection cycles of vacuole-adapted bacteria that target cholesterol trafficking—Anaplasma phagocytophilum, Coxiella burnetii, Chlamydia trachomatis, and Chlamydia pneumoniae. Vacuole maturation, replication, and infectious progeny generation by A. phagocytophilum, which exclusively hijacks LDL cholesterol, are halted and C. burnetii, for which lysosomal cholesterol accumulation is bactericidal, is killed by FIASMAs. Infection cycles of Chlamydiae, which hijack LDL cholesterol and other lipid sources, are suppressed but less so than A. phagocytophilum or C. burnetii. A. phagocytophilum fails to productively infect ASM−/− or FIASMA-treated mice. These findings establish the importance of ASM for infection by intracellular bacteria and identify FIASMAs as potential host-directed therapies for diseases caused by pathogens that manipulate LDL cholesterol.







