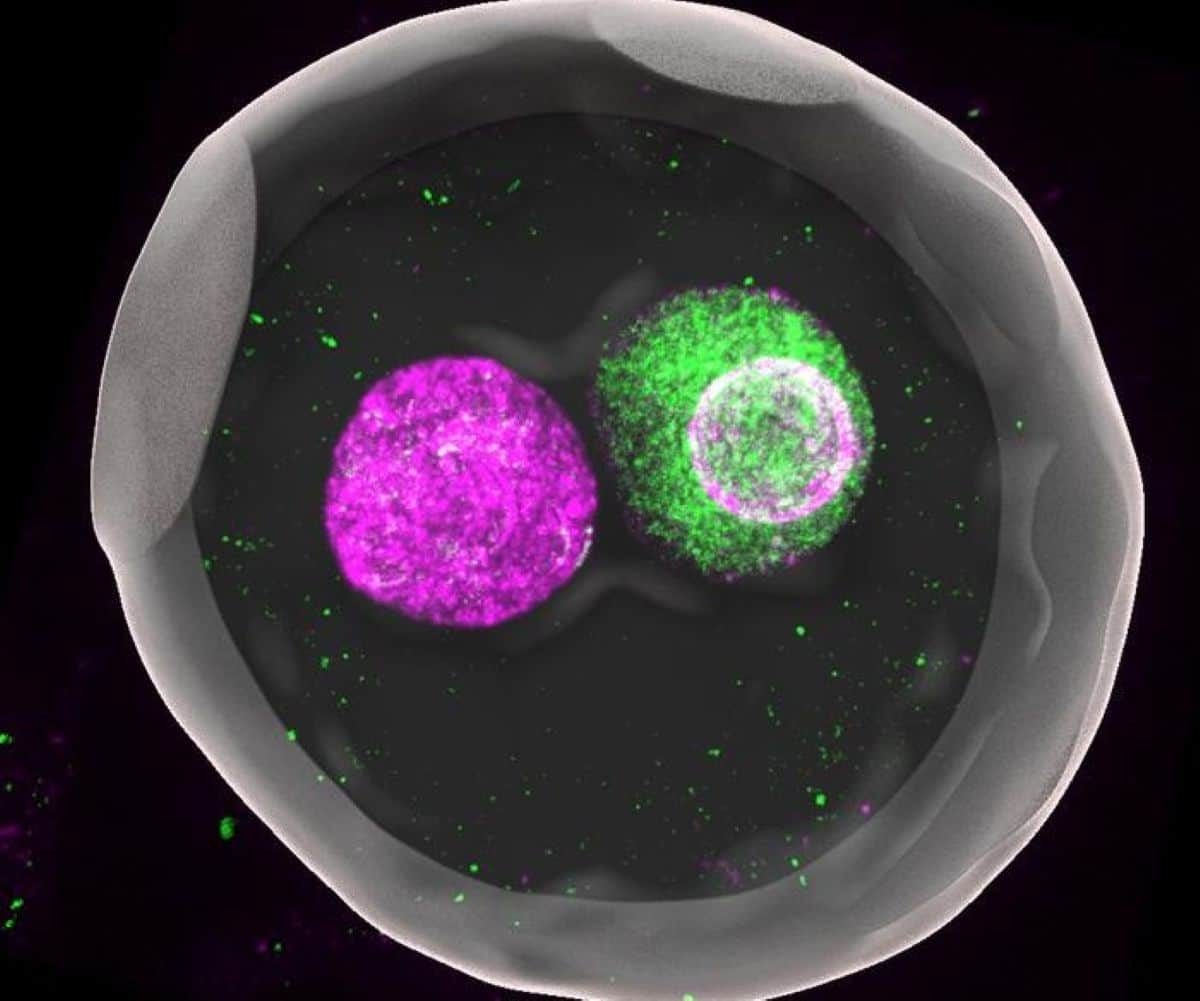
Summary: For decades, biologists observed a strange phenomenon in the first hours of life: after a sperm fertilizes an egg, their nuclei (pronuclei) stay separate instead of fusing immediately. A new study finally explains why. It turns out this separation is a high-stakes “competition.”
By staying apart, the maternal and paternal nuclei race to absorb growth factors, which keeps each nucleus small. This physical constraint is essential for preserving the “chemical tags” (epigenetic modifications) on the genome that allow a zygote to develop into a healthy embryo.
Key Facts
- The Competition Mechanism: The two pronuclei engage in a biological race to absorb growth factors from the surrounding cell fluid. This competition prevents either nucleus from growing too large.
- Size Matters: If the nuclei fuse early (becoming one large nucleus), the competition vanishes. The resulting “super-nucleus” grows too big, which dilutes and destroys the essential chemical tags (histone modifications) required for gene regulation.
- Epigenetic Blueprint: The maternal genome arrives with specific chemical markers that the sperm genome lacks. These tags are the instructions for early development; without them, the embryo fails to develop to term.
- The “Third Nucleus” Experiment: To prove the theory, researchers added a third temporary nucleus to fused embryos. This re-established the competition, limited nuclear size, and partially restored the embryo’s developmental potential.
- Assisted Reproduction Insights: This explains why embryos with early-fused nuclei in IVF settings have lower success rates. It highlights that spatial organization is as critical as genetic information.
Source: Kobe University
At the beginning of a new individual’s life, an egg and a sperm fuse to form what biologists call a “zygote,” a fertilized egg. In mammals such as mice and humans, the DNA-carrying nuclei of these cells don’t fuse right away but remain separate as so-called “pronuclei” until just before the first cell division.
“This fact has been known for decades, but no one really understood why this separation exists. People just treated it as a descriptive feature rather than something to be tested experimentally,” says Kobe University developmental biologist KYOGOKU Hirohisa.
If anything, separate nuclei may increase the risk of errors during the first cell division. In assisted reproduction, embryos in which the maternal and paternal pronuclei are fused early have therefore attracted interest, but these embryos are known to have a lower likelihood of developing to term.
Kyogoku specializes in studying early development directly after fertilization.
“The DNA an individual inherits from their mother and father differs not only in its genetic information. It is also known that the maternal genome has a lot of chemical modifications, like little tags, on the molecules around which the DNA is wrapped. The genome from the sperm, on the other hand, has virtually no such modifications. But these proper modifications are essential for embryonic development,” he explains.
Thus suspecting that the fusion of pronuclei might have an influence on gene regulation, he teamed up with researchers from RIKEN and, using his special cell manipulation techniques, took a close look at what happened to these modifications when the pronuclei are fused and under other conditions.
In the journal Nature, the team now published that they found that the size of the pronuclei is essential for maintaining the regulatory modifications. Whenever the size of the nucleus was large, either through manipulation or because of the premature fusing of the maternal and paternal pronuclei, the degree of the chemical tagging became low.
And when they investigated what keeps the pronuclei’s sizes small in the natural, separated state, they found that the two pronuclei engage in a race to absorb factors that regulate nucleus growth.
This then yielded a clear mechanistic explanation for why the developmental potential of fused nuclei is smaller: Because the single nucleus doesn’t have to compete for these factors it ends up much larger and as a result the genome’s regulatory tags get lost.
“I find it exciting that a very familiar structure — two separate nuclei in a fertilized egg — turns out to have a clear and functional role, that is, it actively creates a competitive environment inside the cell which helps maintain proper regulation and supports development,” says Kyogoku.
To test their theory, the team temporarily introduced into zygotes with prematurely fused pronuclei an additional pronucleus to re-establish competition. And indeed, this intervention successfully limited nuclear size and partially restored the regulatory marks as well as the developmental potential.
The Kobe University work opens a whole new chapter of research into the very first steps a new life takes. “Even at the beginning of life, spatial organization is not just incidental but fundamentally important,” explains Kobe University biologist Kyogoku. He continues, “This result brings us one step closer to understanding the physical and biological principles that explain why early embryos are both robust and error-prone at the same time.”
Funding: This research was funded by the Japan Society for the Promotion of Science (grants JP25H00981, JP23H04947, JP21H02407, JP18H05549, JP20H05376, JP22H04674, JP25H01445, 19K14673, JP20H05368, JP21H05292, JP18H05527, JP20H00456, JP22H04676, 19H05754), the Japan Science and Technology Agency (grants JPMJPR20K4, JPMJPR19K7, JPMJFR2338) and the Japan Agency for Medical Research and Development (grant JP22ama121017j0001). It was conducted in collaboration with researchers from the RIKEN Center for Biosystem Dynamics, Kyushu University, and the RIKEN Center for Integrative Medical Sciences.
Key Questions Answered:
A: They don’t actually “fuse” into a single working nucleus in the way people think. They stay separate right up until the first cell division begins. At that point, the nuclear envelopes break down so the maternal and paternal chromosomes can line up and divide into two new cells. The “separation” is a protective holding pattern.
A: Potentially. Knowing that nuclear size and spatial competition are the “secret sauce” for healthy gene regulation gives embryologists a new metric to monitor. We might eventually develop techniques to ensure this “tug-of-war” stays balanced in lab-grown embryos.
A: They are mostly histone modifications. DNA is wrapped around proteins called histones. The “tags” are small molecules (like methyl or acetyl groups) that tell the cell whether to “read” or “ignore” a specific gene. If the nucleus gets too big, these tags get lost or spread too thin.
Editorial Notes:
- This article was edited by a Neuroscience News editor.
- Journal paper reviewed in full.
- Additional context added by our staff.
About this genetics research news
Author: Daniel Schenz
Source: Kobe University
Contact: Daniel Schenz – Kobe University
Image: The image is credited to KYOGOKU Hirohisa
Original Research: Open access.
“Cytoplasmic competition between separate parental pronuclei in zygotes” by Hirohisa Kyogoku, Mitsusuke Tarama, Masahiro Matsuwaka, Tappei Mishina, Akihito Harada, Reiko Nakagawa, Mami Kumon, Yoshihiro Shimizu, Yasuyuki Ohkawa, Tatsuo Shibata, Azusa Inoue & Tomoya S. Kitajima. Nature
DOI:10.1038/s41586-026-10417-7
Abstract
Cytoplasmic competition between separate parental pronuclei in zygotes
Embryogenesis begins with a zygote—a single cell with two pronuclei that separately enclose maternal and paternal chromosomes. The functional significance of the separation of parental chromosomes into distinct pronuclei remains unexplored, despite the fact that one-pronuclear biparental zygotes are used clinically.
Here, using a combination of mouse zygote manipulation, quantitative imaging and theoretical approaches, we show a cytoplasm-mediated competition mechanism between separate parental pronuclei that ensures developmental potential.
This mechanism limits pronuclear volume and prevents epigenetic mark dysregulation, including loss of trimethylated histones.
One-pronuclear biparental zygotes lack this mechanism, resulting in a reduced rate of development to term. This low developmental potential can be partially rescued by competition-based or drug-based restoration of epigenetic marks.
This study provides a spatial mechanism linking fertilization to the establishment of the full developmental potential for the next generation, highlighting caveats in clinical use of one-pronuclear biparental zygotes.