Summary: Researchers report neurons move the site of microRNA maturation away from the cytoplasm out to the dendrites.
Source: Max Planck Institute.
Witnessing the birth of a tiny RNA at brain synapses.
Proteins are the building blocks of all cells. They are made from messenger RNA (mRNA) molecules, which are copied from DNA in the nuclei of cells. All cells, including brain cells regulate the amount and kind of proteins they make with the help of very small “non-coding” RNAs, so-called microRNAs. Scientists from the Max Planck Institute for Brain Research and Goethe University in Frankfurt, respectively, now show that neurons move the site of microRNA maturation away from the cytoplasm out to the dendrites, thin processes, which are closer to where synapses are. This puts the newly born microRNA into much smaller environment with fewer mRNA target options.
microRNAs prevent the mRNA from being made into protein. They are made from larger precursor RNA molecules by several processing steps in the nucleus and cytoplasm. In individual cells, copy numbers of most microRNAs in single cells are relatively low in contrast to potential mRNA targets within individual cells where copy numbers can be up to 10,000 molecules. As such, the absolute number of potential mRNA targets within a cell for a single microRNA species could be very high (e.g. millions), raising the question of how a microRNA can effectively regulate a particular target mRNA.
Scientists from the lab of Erin Schuman and Alexander Heckel discovered how neurons have solved the abundance problem: microRNAs do not mature in the cytoplasm as is the case in other cells, but rather in the dendrites. “We tested our hypothesis by using a clever design of a fluorescent molecular reporter, modelled after an immature microRNA”, Heckel says. “We filled neurons with this probe and then stimulated individual synapses. To our surprise, we could then see bright fluorescent spots at the stimulated synapses, showing us the birth of the microRNA. We then saw that the microRNA target was downregulated in the neighborhood of the dendrite where the microRNA was born.”
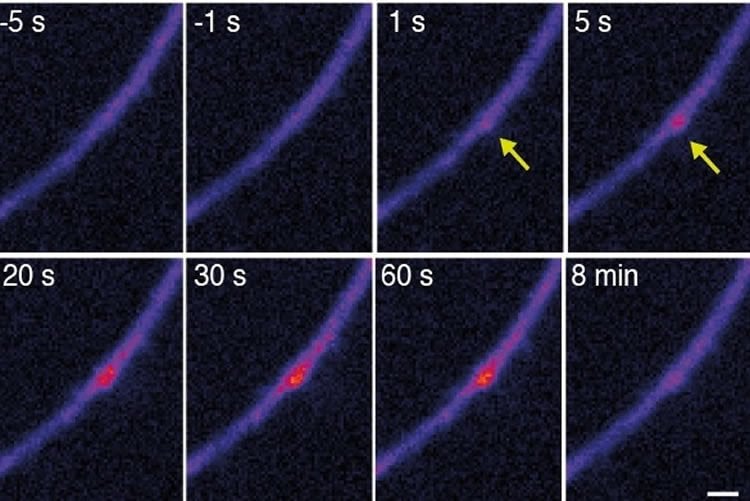
An important feature of neurons is their ability to communicate with one another at synapses, the points of contact between two cells. Synapses use proteins that are synthesized close-by to fuel communication and the formation of memories. Schuman: “By moving the birthplace of the microRNA to the dendrites and synapses where it is closer to its targets, neurons have solved the microRNA-mRNA numbers game and gained a way for external events-resulting in the activation of synapses, to control the local expression of important brain molecules which is important for neuronal communication and also for memory formation.”
Source: Saori Obayashi – Max Planck Institute
Image Source: NeuroscienceNews.com image is credited to Sambandan et al, 2017.
Original Research: Abstract for “Activity-dependent spatially-localized miRNA maturation in neuronal dendrites” by Sambandan, S., Akbalik, G., Kochen, L., Rinne, J. Kahlstatt, J., Glock, C. Tushev, G., Alvarez-Castelao, B., Heckel, A. and Schuman, E.M. in Science. Published online February 10 2017 doi:10.1126/science.aaf8995
[cbtabs][cbtab title=”MLA”]Max Planck Institute “Bright Spots in Brain Cells.” NeuroscienceNews. NeuroscienceNews, 10 February 2017.
<https://neurosciencenews.com/rna-synapses-neuroscience-6095/>.[/cbtab][cbtab title=”APA”]Max Planck Institute (2017, February 10). Bright Spots in Brain Cells. NeuroscienceNew. Retrieved February 10, 2017 from https://neurosciencenews.com/rna-synapses-neuroscience-6095/[/cbtab][cbtab title=”Chicago”]Max Planck Institute “Bright Spots in Brain Cells.” https://neurosciencenews.com/rna-synapses-neuroscience-6095/ (accessed February 10, 2017).[/cbtab][/cbtabs]
Abstract
Activity-dependent spatially-localized miRNA maturation in neuronal dendrites
MicroRNAs (miRNAs) regulate gene expression by binding to target messenger RNAs (mRNAs) and preventing their translation. In general, the number of potential mRNA targets in a cell is much greater than the miRNA copy number, complicating high-fidelity miRNA-target interactions. We developed an inducible fluorescent probe to explore whether the maturation of a miRNA could be regulated in space and time in neurons. A precursor miRNA (pre-miRNA) probe exhibited an activity-dependent increase in fluorescence, suggesting the stimulation of miRNA maturation. Single-synapse stimulation resulted in a local maturation of miRNA that was associated with a spatially restricted reduction in the protein synthesis of a target mRNA. Thus, the spatially and temporally regulated maturation of pre-miRNAs can be used to increase the precision and robustness of miRNA-mediated translational repression.
“Activity-dependent spatially-localized miRNA maturation in neuronal dendrites” by Sambandan, S., Akbalik, G., Kochen, L., Rinne, J. Kahlstatt, J., Glock, C. Tushev, G., Alvarez-Castelao, B., Heckel, A. and Schuman, E.M. in Science. Published online February 10 2017 doi:10.1126/science.aaf8995