Researchers in Japan have revealed the molecular mechanism involved in the process of repair to damage of the myelin sheath.
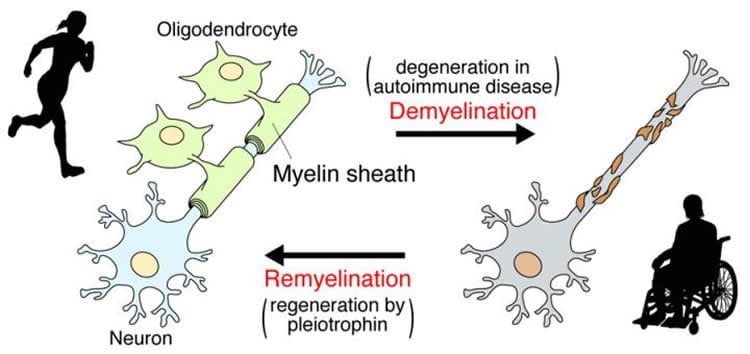
In vertebrates, axons extending from nerve cells are covered by insulating sheets called the myelin sheath, made with the cell membranes of oligodendrocytes, enabling fast electrical signaling through saltatory conduction. Normally, myelin is repaired, even if damaged, but the mechanism that controls remyelination was not well understood. In addition, in demyelinating diseases such as multiple sclerosis, the myelin sheath does not recover from damage and gets worse, finally leading to symptoms such as vision loss, limb numbness, and movement disorders.
The research group of Professor Masaharu Noda and colleagues of the National Institute for Basic Biology, a member institute of the National Institutes of Natural Sciences, performed a detailed examination of the remyelinating process of damaged myelin using disease model mice. Their results show that a growth factor called pleiotrophin is secreted from nerve axons injured by demyelination, and this pleiotrophin inhibits the function of the receptor molecule PTPRZ of oligodendrocyte precursor cells, stimulating cellular differentiation into oligodendrocytes which form the myelin sheath, thereby promoting remyelination.
This achievement shows that it is possible to encourage the regeneration of the myelin sheath by inhibiting the action of PTPRZ in endogenous oligodendrocyte precursor cells, indicating a new potential treatment for demyelinating diseases.
The results of this research were published in The Journal of Neuroscience on September 2, 2015.
Professor Noda said “This achievement was made possible by establishing oligodendrocyte precursor cell lines. Pleiotrophin is an endogenous PTPRZ inhibitor, but if synthetic PTPRZ inhibitors were obtained, then effective treatments for multiple sclerosis should become possible. We are currently directing our research in that direction”.
Source: NIBB’s Office of Public Relations – NIBB
Image Source: The image is credited to NIBB
Original Research: Abstract for “Inactivation of Protein Tyrosine Phosphatase Receptor Type Z by Pleiotrophin Promotes Remyelination through Activation of Differentiation of Oligodendrocyte Precursor Cells” by Kazuya Kuboyama, Akihiro Fujikawa, Ryoko Suzuki, and Masaharu Noda in Journal of Neuroscience. Published online September 2 2015 doi:10.1523/JNEUROSCI.2127-15.2015
Abstract
Inactivation of Protein Tyrosine Phosphatase Receptor Type Z by Pleiotrophin Promotes Remyelination through Activation of Differentiation of Oligodendrocyte Precursor Cells
Multiple sclerosis (MS) is a progressive neurological disorder associated with myelin destruction and neurodegeneration. Oligodendrocyte precursor cells (OPCs) present in demyelinated lesions gradually fail to differentiate properly, so remyelination becomes incomplete. Protein tyrosine phosphatase receptor type Z (PTPRZ), one of the most abundant protein tyrosine phosphatases expressed in OPCs, is known to suppress oligodendrocyte differentiation and maintain their precursor cell stage. In the present study, we examined the in vivo mechanisms for remyelination using a cuprizone-induced demyelination model. Ptprz-deficient and wild-type mice both exhibited severe demyelination and axonal damage in the corpus callosum after cuprizone feeding. The similar accumulation of OPCs was observed in the lesioned area in both mice; however, remyelination was significantly accelerated in Ptprz-deficient mice after the removal of cuprizone. After demyelination, the expression of pleiotrophin (PTN), an inhibitory ligand for PTPRZ, was transiently increased in mouse brains, particularly in the neurons involved, suggesting its role in promoting remyelination by inactivating PTPRZ activity. In support of this view, oligodendrocyte differentiation was augmented in a primary culture of oligodendrocyte-lineage cells from wild-type mice in response to PTN. In contrast, these cells from Ptprz-deficient mice showed higher oligodendrocyte differentiation without PTN and differentiation was not enhanced by its addition. We further demonstrated that PTN treatment increased the tyrosine phosphorylation of p190 RhoGAP, a PTPRZ substrate, using an established line of OPCs. Therefore, PTPRZ inactivation in OPCs by PTN, which is secreted from demyelinated axons, may be the mechanism responsible for oligodendrocyte differentiation during reparative remyelination in the CNS.
SIGNIFICANCE STATEMENT Multiple sclerosis (MS) is an inflammatory disease of the CNS that destroys myelin, the insulation that surrounds axons. Associated damages to oligodendrocytes (the cells that produce myelin) and nerve fibers produce neurological disability. Most patients with MS have an initial relapsing-remitting course for 5–15 years. Remyelination during the early stages of the disease process has been documented; however, the molecular mechanism underlying remyelination has not been understood. Protein tyrosine phosphatase receptor type Z (PTPRZ) is a receptor-like protein tyrosine phosphatase preferentially expressed in the CNS. This study shows that pleiotrophin, an inhibitory ligand for PTPRZ, is transiently expressed and released from demyelinated neurons to inactivate PTPRZ in oligodendrocyte precursor cells present in the lesioned part, thereby allowing their differentiation for remyelination.
“Inactivation of Protein Tyrosine Phosphatase Receptor Type Z by Pleiotrophin Promotes Remyelination through Activation of Differentiation of Oligodendrocyte Precursor Cells” by Kazuya Kuboyama, Akihiro Fujikawa, Ryoko Suzuki, and Masaharu Noda in Journal of Neuroscience. Published online September 2 2015 doi:10.1523/JNEUROSCI.2127-15.2015







