Summary: Researchers successfully used a synthetic compound to stimulate a receptor pathway to promote remyelination in the brain. The technique may have significant beneficial implications for treating multiple sclerosis, researchers say.
Source: University of Melbourne.
University of Melbourne researchers have found a way to rebuild damaged nerve coverings that cause Multiple Sclerosis.
Finding ways to restore the myelin sheath is recognised as important to preventing the progression of disability in MS patients.
Researcher Jessica Fletcher led the team who made the discovery and their findings are published in The Journal of Neuroscience.
“Your brain runs on electricity. And, like electrical wires, your nervous system needs insulation. These nerves are covered by an insulating sheath called myelin that is vital to the normal functioning of our nervous system,” Dr Fletcher said.
“But for those people affected by diseases like MS, this insulating myelin is destroyed by the immune system – leading to significant nerve dysfunction as well as slowed or blocked nerve conduction between the brain and the rest of the body.”
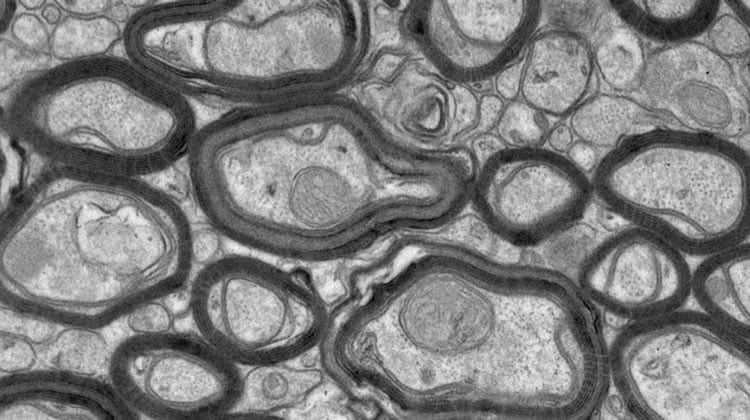
Dr Fletcher said the team successfully used a synthetic compound to stimulate a receptor pathway to promote remyelination in the brain.
“There’s nothing currently available to help with myelin sheath repair. The beauty of what our team has done is taken what naturally occurs in healthy cells and used that to manipulate a similar response in damaged cells,” she said.
“It’s very basic foundation research to show that this idea can work.”
Dr Fletcher said this was early-stage research and any medical application to the discovery would be a long way off.
Source: Kathryn Powley – University of Melbourne
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Dr David Gonsalvez/University of Melbourne.
Original Research: Abstract for “Targeting TrkB with a Brain-Derived Neurotrophic Factor Mimetic Promotes Myelin Repair in the Brain” by Jessica L. Fletcher, Rhiannon J. Wood, Jacqueline Nguyen, Eleanor M.L. Norman, Christine M.K. Jun, Alexa R. Prawdiuk, Melissa Biemond, Huynh T.H. Nguyen, Susan E. Northfield, Richard A. Hughes, David G. Gonsalvez, Junhua Xiao and Simon S. Murray Journal of Neuroscience. Published August 8 2018.
doi:10.1523/JNEUROSCI.0487-18.2018
[cbtabs][cbtab title=”MLA”]University of Melbourne”Possible New Ways to Regenerate Myelin.” NeuroscienceNews. NeuroscienceNews, 27 August 2018.
<https://neurosciencenews.com/myelin-regeneration-9749/>.[/cbtab][cbtab title=”APA”]University of Melbourne(2018, August 27). Possible New Ways to Regenerate Myelin. NeuroscienceNews. Retrieved August 27, 2018 from https://neurosciencenews.com/myelin-regeneration-9749/[/cbtab][cbtab title=”Chicago”]University of Melbourne”Possible New Ways to Regenerate Myelin.” https://neurosciencenews.com/myelin-regeneration-9749/ (accessed August 27, 2018).[/cbtab][/cbtabs]
Abstract
Targeting TrkB with a Brain-Derived Neurotrophic Factor Mimetic Promotes Myelin Repair in the Brain
Methods to promote myelin regeneration in response to central myelin loss are essential to prevent the progression of clinical disability in demyelinating diseases. The neurotrophin brain-derived neurotrophic factor (BDNF) is known to promote myelination during development via oligodendrocyte expressed TrkB receptors. Here, we use a structural mimetic of BDNF to promote myelin regeneration in a preclinical mouse model of central demyelination. In female mice, we show that selective targeting of TrkB with the BDNF-mimetic enhances remyelination, increasing oligodendrocyte differentiation, the frequency of myelinated axons, and myelin sheath thickness after a demyelinating insult. Treatment with exogenous BDNF exerted an attenuated effect, increasing myelin sheath thickness only. Further, following conditional deletion of TrkB from premyelinating oligodendrocytes, we show the effects of the BDNF-mimetic on oligodendrocyte differentiation and remyelination are lost, indicating these are dependent on oligodendrocyte expression of TrkB. Overall, these studies demonstrate that targeting oligodendrocyte TrkB promotes in vivo remyelination in the brain.
SIGNIFICANCE STATEMENT
Novel strategies to promote myelin regeneration are required to prevent progressive neurodegeneration and clinical disability in patients with central demyelinating disease. Here, we test whether selectively targeting the TrkB receptor on the myelin-producing oligodendrocytes, can promote remyelination in the brain. Using a structural mimetic of its native ligand, BDNF, we show that stimulation of TrkB enhances remyelination, increasing oligodendrocyte differentiation, the frequency of myelinated axons and thickness of the myelin sheath following a demyelinating insult. Further, we show that these effects are dependent on the phosphorylation of oligodendrocyte expressed TrkB receptors in vivo. Overall, we demonstrate that selective targeting of TrkB has therapeutic potential to promote remyelination in the brain.







