Summary: Researchers report a collapse of the mitochondria-associated membrane is a common halmark in two genetic forms of ALS.
Source: Nagoya University.
Amyotrophic lateral sclerosis (ALS) is an adult onset, fetal neurodegenerative disease that selectively affects motor neurons. To date, more than 20 genes are identified as a causative of inherited ALS, and many researchers investigate the pathomechanism of ALS.
A research team led by Prof. Koji Yamanaka (Nagoya University) found that collapse of the MAM is a common pathological hallmark to two distinct inherited forms of ALS: SOD1- and SIGMAR1- linked ALS. The research findings were reported in EMBO Molecular Medicine on November 7th, 2016.
The researchers focused on the mitochondria-associated membrane (MAM), which is a contacting site of mitochondria and endoplasmic reticulum (ER). Recent studies have revealed that the MAM plays a key role in cellular homeostasis, such as lipid synthesis, protein degradation, and energy metabolism. Intriguingly, a recessive mutation in SIGMAR1 gene, which encodes sigma 1 receptor (Sig1R), a chaperone enriched in the MAM, is causative for a juvenile ALS. In this study, the researchers identified a novel ALS-linked SIGMAR1 mutation, c.283dupC/p.L95fs in a juvenile-onset ALS case. Moreover, ALS-linked Sig1R mutant proteins were unstable and non-functional, indicating a loss-of function mechanism in SIGMAR1-linked ALS.
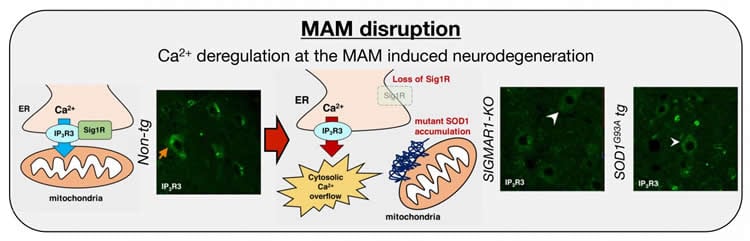
A loss of Sig1R function induced MAM disruption in neurons. However, it was still unknown whether the MAM alternation was also involved in the other ALS cases. To address this question, the researchers cross-bred SIGMAR1 deficient mice with the other inherited ALS mice which overexpress a mutant form of SOD1 gene. SIGMAR1 deficiency accelerated disease onset of SOD1-ALS mice by more than 20 %. In those mice, inositol triphosphate receptor type-3 (IP3R3), a MAM-enriched calcium ion (Ca2+) channel on ER, was disappeared from the MAM. The loss of proper localization of IP3R3 led to Ca2++ dysregulation to exacerbate the neurodegeneration. The researchers also found that IP3R3 was selectively enriched in motor neurons, suggesting that integrity of the MAM is crucial for the selective vulnerability in ALS.
These results provide us with new perspectives regarding future therapeutics, especially focused on preventing the MAM disruption for ALS patients. Together with the research from other groups, collapse of the MAM is widely observed in the other genetic causes of ALS, and therefore it may be applicable to sporadic ALS patients.
Funding: The study was funded by Ministry for Education, Culture and Sports, Science and Technology, Japan Agency for Medical Research and Development, Naito Foundation, Uehara Memorial Foundation, Japan ALS Association, Hori Science and Arts Foundation.
Source: Koomi Sung – Nagoya University
Image Source: This NeuroscienceNews.com image is credited to Koji Yamanaka laboratory.
Original Research: Full open access research for “Mitochondria‐associated membrane collapse is a common pathomechanism in SIGMAR1‐ and SOD1‐linked ALS” by Seiji Watanabe, Hristelina Ilieva, Hiromi Tamada, Hanae Nomura, Okiru Komine, Fumito Endo, Shijie Jin, Pedro Mancias, Hiroshi Kiyama, and Koji Yamanaka in EMBO Molecular Medicine. Published online November 7 2016 doi:10.15252/emmm.201606403
[cbtabs][cbtab title=”MLA”]Nagoya University. “Collapse of Mitochondria-Associated Membrane in ALS.” NeuroscienceNews. NeuroscienceNews, 8 November 2016.
<https://neurosciencenews.com/mitochondria-als-5456/>.[/cbtab][cbtab title=”APA”]Nagoya University. (2016, November 8). Collapse of Mitochondria-Associated Membrane in ALS. NeuroscienceNews. Retrieved November 8, 2016 from https://neurosciencenews.com/mitochondria-als-5456/[/cbtab][cbtab title=”Chicago”]Nagoya University. “Collapse of Mitochondria-Associated Membrane in ALS.” https://neurosciencenews.com/mitochondria-als-5456/ (accessed November 8, 2016).[/cbtab][/cbtabs]
Abstract
Mitochondria‐associated membrane collapse is a common pathomechanism in SIGMAR1‐ and SOD1‐linked ALS
A homozygous mutation in the gene for sigma 1 receptor (Sig1R) is a cause of inherited juvenile amyotrophic lateral sclerosis (ALS16). Sig1R localizes to the mitochondria‐associated membrane (MAM), which is an interface of mitochondria and endoplasmic reticulum. However, the role of the MAM in ALS is not fully elucidated. Here, we identified a homozygous p.L95fs mutation of Sig1R as a novel cause of ALS16. ALS‐linked Sig1R variants were unstable and incapable of binding to inositol 1,4,5‐triphosphate receptor type 3 (IP3R3). The onset of mutant Cu/Zn superoxide dismutase (SOD1)‐mediated ALS disease in mice was accelerated when Sig1R was deficient. Moreover, either deficiency of Sig1R or accumulation of mutant SOD1 induced MAM disruption, resulting in mislocalization of IP3R3 from the MAM, calpain activation, and mitochondrial dysfunction. Our findings indicate that a loss of Sig1R function is causative for ALS16, and collapse of the MAM is a common pathomechanism in both Sig1R‐ and SOD1‐linked ALS. Furthermore, our discovery of the selective enrichment of IP3R3 in motor neurons suggests that integrity of the MAM is crucial for the selective vulnerability in ALS.
“Mitochondria‐associated membrane collapse is a common pathomechanism in SIGMAR1‐ and SOD1‐linked ALS” by Seiji Watanabe, Hristelina Ilieva, Hiromi Tamada, Hanae Nomura, Okiru Komine, Fumito Endo, Shijie Jin, Pedro Mancias, Hiroshi Kiyama, and Koji Yamanaka in EMBO Molecular Medicine. Published online November 7 2016 doi:10.15252/emmm.201606403






