Summary: Iron levels in the basal ganglia steadily increase throughout development and, in two sub-regions, continue to increase into adulthood. Decreased levels of iron in the putamen was correlated with impaired cognitive performance involving reasoning and spatial processing. Findings suggest the brain requires iron for healthy cognitive development.
Source: SfN
Iron levels in brain tissue rise during development and are correlated with cognitive abilities, according to research in children and young adults recently published in Journal of Neuroscience. Future work could lead to iron supplementation as an intervention for atypical cognitive development.
Brain cells stay healthy in part by storing iron. It is most concentrated in the basal ganglia, a brain region that filters incoming information from moment to moment and suggests the best action to take. Low iron in the basal ganglia during early life is linked to cognitive impairment, yet we don’t know how iron levels change during typical development.
Larsen et al. examined brain iron levels through magnetic resonance imaging brain scans from the Philadelphia Neurodevelopmental Cohort, a dataset of scans from over 1500 children and young adults ranging from age eight to twenty-four. The research team found that brain iron levels in the basal ganglia steadily increase throughout development and, in two subregions, continue to increase into adulthood.
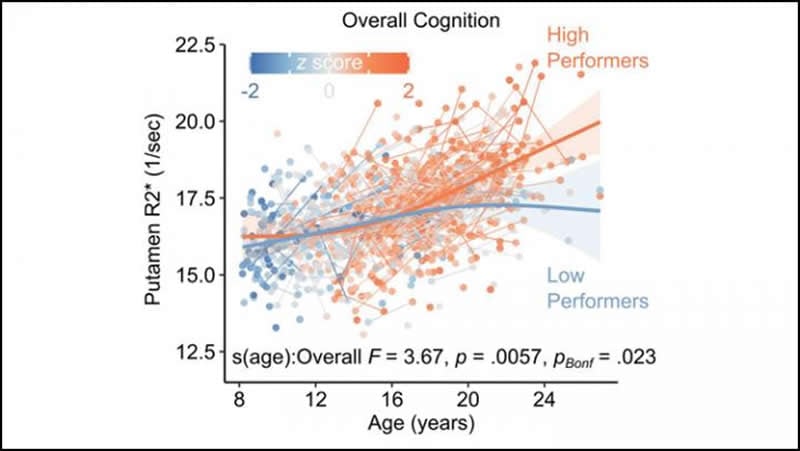
Decreased brain iron in one subregion, the putamen, was correlated with impaired performance on cognitive tasks involving reasoning and spatial processing, suggesting that brain iron is needed for healthy cognitive development.
Source:
SfN
Media Contacts:
Calli McMurray – SfN
Image Source:
The image is credited to Larsen et al., JNeurosci 2020.
Original Research: Closed access
“Longitudinal Development of Brain Iron Is Linked to Cognition in Youth”. Bart Larsen, Josiane Bourque, Tyler M. Moore, Azeez Adebimpe, Monica E. Calkins, Mark A. Elliott, Ruben C. Gur, Raquel E. Gur, Paul J. Moberg, David R. Roalf, Kosha Ruparel, Bruce I. Turetsky, Simon N. Vandekar, Daniel H. Wolf, Russell T. Shinohara and Theodore D. Satterthwaite.
Journal of Neuroscience doi:10.1523/JNEUROSCI.2434-19.2020.
Abstract
Longitudinal Development of Brain Iron Is Linked to Cognition in Youth
Brain iron is vital to multiple aspects of brain function, including oxidative metabolism, myelination, and neurotransmitter synthesis. Atypical iron concentration in the basal ganglia is associated with neurodegenerative disorders in aging and cognitive deficits. However, the normative development of brain iron concentration in adolescence and its relationship to cognition are less well understood. Here, we address this gap in a longitudinal sample of 922 humans aged 8-26 years at the first visit (M = 15.1, SD = 3.72; 336 males, 486 females) with up to four multi-echo T2* scans each. Using this sample of 1,236 imaging sessions, we assessed the longitudinal developmental trajectories of tissue iron in the basal ganglia. We quantified tissue iron concentration using R2* relaxometry within four basal ganglia regions, including the caudate, putamen, nucleus accumbens, and globus pallidus. The longitudinal development of R2* was modeled using generalized additive mixed models (GAMMs) with splines to capture linear and nonlinear developmental processes. We observed significant increases in R2* across all regions, with the greatest and most prolonged increases occurring in the globus pallidus and putamen. Further, we found that the developmental trajectory of R2* in the putamen is significantly related to individual differences in cognitive ability, such that greater cognitive ability is increasingly associated with greater iron concentration through late adolescence and young-adulthood. Together, our results suggest a prolonged period of basal ganglia iron enrichment that extends into the mid-twenties, with diminished iron concentration associated with poorer cognitive ability during late adolescence.
SIGNIFICANCE STATEMENT
Brain tissue iron is essential to healthy brain function. Atypical basal ganglia tissue iron levels have been linked to impaired cognition in iron deficient children and adults with neurodegenerative disorders. However, the normative developmental trajectory of basal ganglia iron concentration during adolescence and its association with cognition are less well understood. In the largest study of tissue iron development yet reported, we characterize the developmental trajectory of tissue iron concentration across the basal ganglia during adolescence and provide evidence that diminished iron content is associated with poorer cognitive performance even in healthy youth. These results highlight the transition from adolescence to adulthood as a period of dynamic maturation of tissue iron concentration in the basal ganglia.






