Researchers in Israel have manipulated the mouse immune system to target Alzheimer’s disease. The research was published in the journal Nature Communications on 18 August 2015.
The research team explored the complex interplay between the body’s immune system and the brain. They used mice that have excess amyloid – a hallmark protein in Alzheimer’s disease – to look at the different roles of the immune system in the disease. They were particularly interested in a special type of cell called regulatory T-cells – white blood cells that keep the immune system in check. Too many of this type of cell can prevent the body’s immune system from responding to damage in the brain.
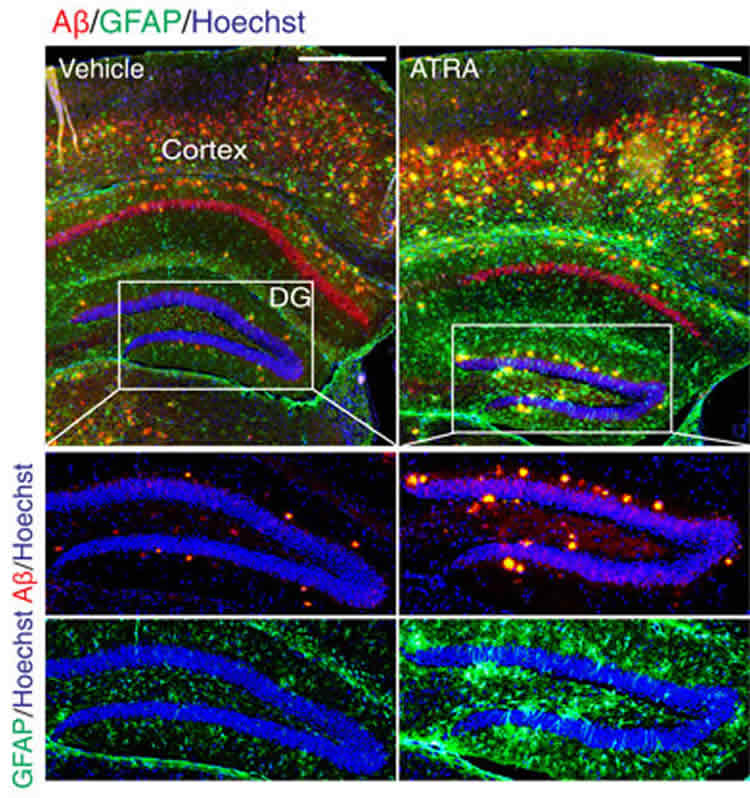
The researchers used two approaches to reduce the number of regulatory T-cells in mice with hallmark features of Alzheimer’s disease. They found that reducing their levels using either a genetic trick or chemical compounds could boost the immune response to nerve cell damage in the brain. They also found that levels of the toxic amyloid protein dropped. This increase in ‘good’ inflammation was also associated with improved memory and thinking skills in the mice. In order to explore the role of regulatory T-cells further, the team took the opposite approach to their earlier experiment and used a compound that boosted their ranks. This led to a decrease in ‘good’ inflammation, an increase in amyloid build-up and a deterioration in memory skills.
Dr Laura Phipps from Alzheimer’s Research UK, said:
“The role of the immune system in Alzheimer’s disease is incredibly complex and researchers are working hard to unravel how best to target it to promote better brain health. This study in mice suggests that boosting immune cells that are entering the brain from the rest of the body could slow Alzheimer’s changes and benefit memory, but it is too soon to know the relevance for people living with dementia. We know that testing lots of different scientific approaches is crucial in the hunt for new treatments and it’s only through increased and sustained investment in dementia research that we will find the answers to our biggest medical challenge.”
Source: Alzheimer’s Research UK
Image Source: The image is credited to the researchers/Nature Communications and is licensed Creative Commons Attribution 4.0 International
Original Research: Full open access research for “Breaking immune tolerance by targeting Foxp3+ regulatory T cells mitigates Alzheimer’s disease pathology” by Kuti Baruch, Neta Rosenzweig, Alexander Kertser, Aleksandra Deczkowska, Alaa Mohammad Sharif, Amit Spinrad, Afroditi Tsitsou-Kampeli, Ayelet Sarel, Liora Cahalon, Michal Schwartz in Nature Communications. Published online August 18 2015 doi:10.1038/ncomms8967
Abstract
Breaking immune tolerance by targeting Foxp3+ regulatory T cells mitigates Alzheimer’s disease pathology
Alzheimer’s disease (AD) is a neurodegenerative disorder in which chronic neuroinflammation contributes to disease escalation. Nevertheless, while immunosuppressive drugs have repeatedly failed in treating this disease, recruitment of myeloid cells to the CNS was shown to play a reparative role in animal models. Here we show, using the 5XFAD AD mouse model, that transient depletion of Foxp3+ regulatory T cells (Tregs), or pharmacological inhibition of their activity, is followed by amyloid-β plaque clearance, mitigation of the neuroinflammatory response and reversal of cognitive decline. We further show that transient Treg depletion affects the brain’s choroid plexus, a selective gateway for immune cell trafficking to the CNS, and is associated with subsequent recruitment of immunoregulatory cells, including monocyte-derived macrophages and Tregs, to cerebral sites of plaque pathology. Our findings suggest targeting Treg-mediated systemic immunosuppression for treating AD.
“Breaking immune tolerance by targeting Foxp3+ regulatory T cells mitigates Alzheimer’s disease pathology” by Kuti Baruch, Neta Rosenzweig, Alexander Kertser, Aleksandra Deczkowska, Alaa Mohammad Sharif, Amit Spinrad, Afroditi Tsitsou-Kampeli, Ayelet Sarel, Liora Cahalon, Michal Schwartz in Nature Communications. Published online August 18 2015 doi:10.1038/ncomms8967






