Summary: Findings could help to find treatments for fight obesity, diabetes and cardiovascular disease, researchers report.
Source: McGill University.
McGill-led discovery could help fight obesity, metabolic disorders.
Researchers have uncovered a new molecular pathway for stimulating the body to burn fat – a discovery that could help fight obesity, diabetes and cardiovascular disease.
In a study published in the journal Genes & Development, a team led by researchers from the Department of Biochemistry at McGill University focus on a protein known as folliculin and its role in regulating the activity of fat cells. By knocking out the gene that produces folliculin in fat cells in mice, the researchers triggered a series of biomolecular signals that switched the cells from storing fat to burning it.
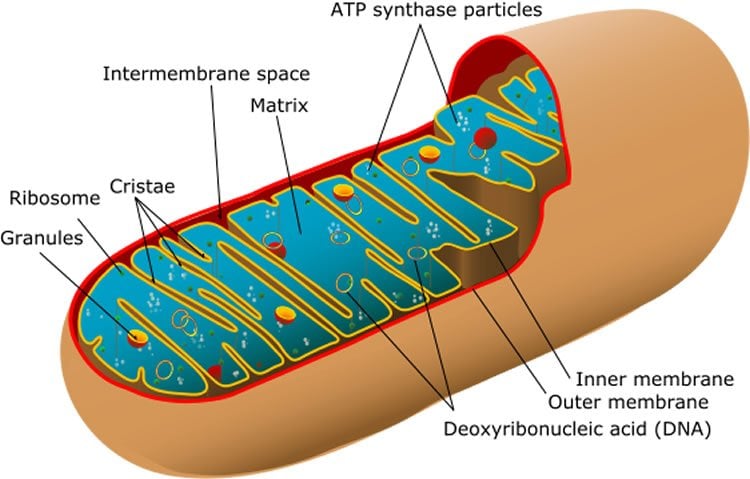
This process is known as the ‘browning’ of fat cells. Brown fat gets its colour from iron-rich mitochondria, an abundance of which is a sign that a cell is in metabolic overdrive. The principal role of brown fat is to burn energy to produce heat, which helps keep our body temperature constant. White fat serves as an energy-storage tissue.
Scientists recently discovered a new type of fat tissue with characteristics somewhere between healthy brown fat and the not-so-healthy white kind. So-called beige fat is capable of behaving like brown fat in response to certain stimuli such as exposure to cold. The more active these cells are, the less likely we are to accumulate unhealthy fat deposits that lead to obesity. Since the discovery of beige fat, the challenge has been to find ways to convert white fat cells into energy-burning beige ones.
“Conversion from white fat cells to beige or brown fat cells is a very desirable effect in the obesity, diabetes, and metabolic syndrome indications, since excess energy in the body is not stored in fat tissue but is burned in brown or beige fat tissue,” said professor Arnim Pause, the study’s senior author.
In collaboration with Vincent Giguère, the team bred mice to have fat cells that did not produce folliculin. They then fed normal mice and folliculin-deficient mice with a high-fat, junk food-like diet over 14 weeks. Normal mice gained weight rapidly, whereas folliculin-deficient mice remained slim and did not suffer the same elevated insulin and triglyceride levels. By measuring rates of oxygen consumption and CO2 production, the researchers found the folliculin-deficient mice were burning more fat. At the end of the trial, these mice had smaller white fat cells and less white fat tissue overall. The extra energy they were producing made them better at tolerating cold temperatures, too.
The breakthrough builds on existing knowledge about two key proteins – PGC-1α and ERRα – and their involvement in regulating mitochondria in fat cells. The researchers, led by graduate student Ming Yan, and Etienne Audet-Walsh, a postdoctoral fellow, found that removing folliculin gives the enzyme known as AMPK free rein to activate these proteins, boosting the number and work rate of the mitochondria in the fat cell. The result is a metabolic reprogramming of fat tissue, turning cells from fat storage units into fat burning engines.
By pinpointing the folliculin pathway, the work has potentially opened the way for new medications to be developed that will stimulate the ‘browning’ process.
“Since that mechanism involves a class of proteins that can be targeted by drugs that are readily absorbed in the body, one implication is that a drug could be developed to stimulate the activity of beige/brown fat cells and thus help manage obesity and other metabolic disorders,” Giguère said.
Funding: Funding for the research was provide by the Canadian Institutes of Health Research, the Fonds de Recherche du Québec-Santé (FRQS), the Kidney Foundation of Canada, the Myrovlytis Trust, a Terry Fox Research Institute Program Project Team Grant and the Canada Foundation for Innovation.
Source: Cynthia Lee – McGill University
Image Source: This NeuroscienceNews.com image is in the public domain.
Original Research: Abstract for “Chronic AMPK activation via loss of FLCN induces functional beige adipose tissue through PGC-1α/ERRα” by Ming Yan, Étienne Audet-Walsh, Sanaz Manteghi, Catherine Rosa Dufour, Benjamin Walker, Masaya Baba, Julie St-Pierre, Vincent Giguère, and Arnim Pause in Genes and Development. Published online May 1 2016 doi:10.1101/gad.281410.116
[cbtabs][cbtab title=”MLA”]McGill University. “Converting Cells to Burn, Not Store, Fat: Mouse Study.” NeuroscienceNews. NeuroscienceNews, 16 May 2016.
<https://neurosciencenews.com/genetics-fat-cells-obesity-4249/>.[/cbtab][cbtab title=”APA”]McGill University. (2016, May 16). Converting Cells to Burn, Not Store, Fat: Mouse Study. NeuroscienceNews. Retrieved May 16, 2016 from https://neurosciencenews.com/genetics-fat-cells-obesity-4249/[/cbtab][cbtab title=”Chicago”]McGill University. “Converting Cells to Burn, Not Store, Fat: Mouse Study.” NeuroscienceNews.
https://neurosciencenews.com/genetics-fat-cells-obesity-4249/ (accessed May 16, 2016).[/cbtab][/cbtabs]
Abstract
Chronic AMPK activation via loss of FLCN induces functional beige adipose tissue through PGC-1α/ERRα
The tumor suppressor folliculin (FLCN) forms a repressor complex with AMP-activated protein kinase (AMPK). Given that AMPK is a master regulator of cellular energy homeostasis, we generated an adipose-specific Flcn (Adipoq-FLCN) knockout mouse model to investigate the role of FLCN in energy metabolism. We show that loss of FLCN results in a complete metabolic reprogramming of adipose tissues, resulting in enhanced oxidative metabolism. Adipoq-FLCN knockout mice exhibit increased energy expenditure and are protected from high-fat diet (HFD)-induced obesity. Importantly, FLCN ablation leads to chronic hyperactivation of AMPK, which in turns induces and activates two key transcriptional regulators of cellular metabolism, proliferator-activated receptor γ (PPARγ) coactivator-1α (PGC-1α) and estrogen-related receptor α (ERRα). Together, the AMPK/PGC-1α/ERRα molecular axis positively modulates the expression of metabolic genes to promote mitochondrial biogenesis and activity. In addition, mitochondrial uncoupling proteins as well as other markers of brown fat are up-regulated in both white and brown FLCN-null adipose tissues, underlying the increased resistance of Adipoq-FLCN knockout mice to cold exposure. These findings identify a key role of FLCN as a negative regulator of mitochondrial function and identify a novel molecular pathway involved in the browning of white adipocytes and the activity of brown fat.
“Chronic AMPK activation via loss of FLCN induces functional beige adipose tissue through PGC-1α/ERRα” by Ming Yan, Étienne Audet-Walsh, Sanaz Manteghi, Catherine Rosa Dufour, Benjamin Walker, Masaya Baba, Julie St-Pierre, Vincent Giguère, and Arnim Pause in Genes and Development. Published online May 1 2016 doi:10.1101/gad.281410.116