Summary: A new Zebra fish study reveals a lack of sensory stimulation following birth can impact neurogenesis and brain development.
Source: University of Toronto.
New research by University of Toronto (U of T) neuroscientists sheds light on links between early brain growth and sensations experienced by animals soon after birth.
The findings suggest mobility restrictions or insufficient sensory stimuli impact the production of new brain cells – known as neurogenesis – and brain growth. The likely outcome is that learning and cognition are negatively affected.
“We have identified a form of neural feedback in zebrafish that could link development of the body with that of the brain,” said Zachary Hall, postdoctoral fellow working with developmental neurobiologist Vincent Tropepe in the Department of Cell & Systems Biology in the Faculty of Arts & Science at U of T, and lead author of two studies examining sensory experience-dependent brain growth.
“By testing whether early deficits in sensory experience similarly restrict human brain growth, our findings offer a novel approach to combatting such deficits to maintain normal brain development.”
Sensory experiences of newborns are critical for their developing brains as they help with the maturation of existing neural circuits. However, whether these sensory experiences also impact early neurogenesis has not been clear before now.
In a study published in the Journal of Neuroscience, the researchers reared zebrafish larvae in environments with reduced lighting, limiting their exposure to visual stimulation.
While they found that a dim environment does not affect body or eye growth, it does negatively impact the ability of neurons in the brain generated during the treatment period to survive. As a result, the animal is left with reduced neural capacity, presumably limiting its visual processing abilities.
In a parallel study published in eLife, the researchers manipulated mobility in various ways, producing differing outcomes. By reducing the size of the accessible environment, they saw decreased physical movement that resulted in a decrease in the number of cells from which new brain cells are created – known as progenitor cells. Conversely, when they imposed a current against which the fish were compelled to swim, the amount of progenitor cells increased.
“Interestingly, the experiments also showed that the sensation of movement, detected by sensory cells, increased the production of brain cells,” said Hall. “Blocking the development of these sensory cells prevented the rise in progenitor cells seen in the fish who swam more. However, activating these same cells in immobilised larvae increased the number of progenitor cells, even though the larvae could not move.
“This suggests that measures to simulate increased physical movement in young children could be used to combat abnormal neurogenesis during early brain growth.”
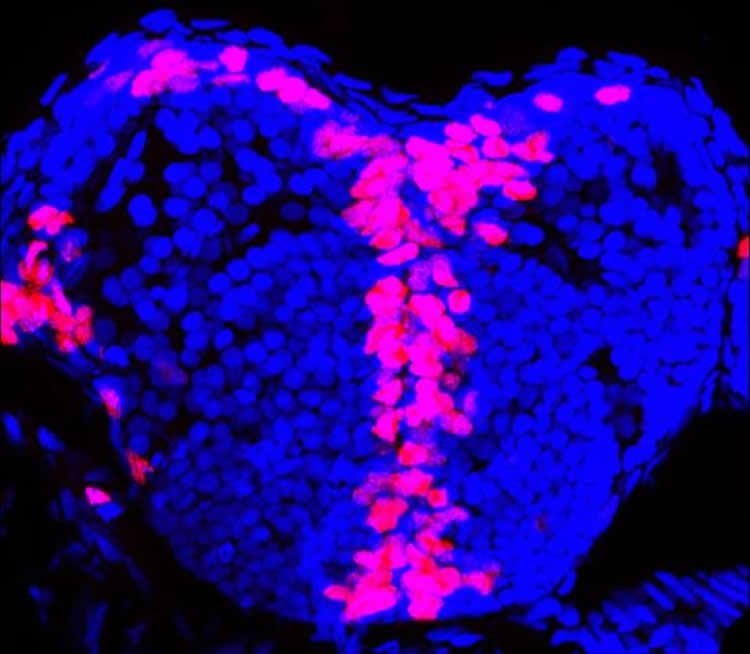
These findings might help explain a number of important links. Doctors in neonatal intensive care units often use massage therapy for babies born prematurely or with illnesses, which seems to be helpful for reasons not fully understood. The researchers suggest sensory feedback might be stimulating neurogenesis that in turn faciliates brain growth.
In contrast, babies born with muscular dystrophy often show signs of impairment in learning and cognitive development. As the disease itself is localized to skeletal muscles, the causes of these behavioural deficiencies are mysterious. Knowing exactly how sensory experiences promote neurogenesis could open the door to post-natal interventions that ensure the development of neurons in the absence of triggers that occur under typical circumstances.
“It’s important to remember that while tantalizing, these links are speculative,” said Tropepe. “Our findings are new and it takes time to truly understand their relevance. Nonetheless, we are excited to learn more about the fundamental nature of sensory-dependent brain growth and its potential medical applications.”
Funding: Support for the research was provided by the Natural Sciences and Engineering Research Council of Canada.
Source: Sean Bettam – University of Toronto
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Zachary Hall/Univesity of Toronto.
Original Research: Full open access research for “Movement maintains forebrain neurogenesis via peripheral neural feedback in larval zebrafish” by Zachary Jonas Hall and Vincent Tropepe Is a corresponding author in eLife. Published March 12 2018.
doi:10.7554/eLife.31045
Abstract for “Visual Experience Facilitates BDNF-Dependent Adaptive Recruitment of New Neurons in the Postembryonic Optic Tectum” by Zachary Jonas Hall and Vincent Tropepe Is a corresponding author in Journal of Neuroscience. Published February 21 2018.
doi:10.1523/JNEUROSCI.1962-17.2018
[cbtabs][cbtab title=”MLA”]University of Toronto “Connection Between Postnatal Sensory Experiences and Brain Development.” NeuroscienceNews. NeuroscienceNews, 16 April 2018.
<https://neurosciencenews.com/brain-development-postnatal-sensation-8808/>.[/cbtab][cbtab title=”APA”]University of Toronto (2018, April 16). Connection Between Postnatal Sensory Experiences and Brain Development. NeuroscienceNews. Retrieved April 16, 2018 from https://neurosciencenews.com/brain-development-postnatal-sensation-8808/[/cbtab][cbtab title=”Chicago”]University of Toronto “Connection Between Postnatal Sensory Experiences and Brain Development.” https://neurosciencenews.com/brain-development-postnatal-sensation-8808/ (accessed April 16, 2018).[/cbtab][/cbtabs]
Abstract
Movement maintains forebrain neurogenesis via peripheral neural feedback in larval zebrafish
The postembryonic brain exhibits experience-dependent development, in which sensory experience guides normal brain growth. This neuroplasticity is thought to occur primarily through structural and functional changes in pre-existing neurons. Whether neurogenesis also mediates the effects of experience on brain growth is unclear. Here, we characterized the importance of motor experience on postembryonic neurogenesis in larval zebrafish. We found that movement maintains an expanded pool of forebrain neural precursors by promoting progenitor self-renewal over the production of neurons. Physical cues associated with swimming (bodily movement) increase neurogenesis and these cues appear to be conveyed by dorsal root ganglia (DRG) in the zebrafish body: DRG-deficient larvae exhibit attenuated neurogenic responses to movement and targeted photoactivation of DRG in immobilized larvae expands the pallial pool of proliferative cells. Our results demonstrate the importance of movement in neurogenic brain growth and reveal a fundamental sensorimotor association that may couple early motor and brain development.
Abstract
Visual Experience Facilitates BDNF-Dependent Adaptive Recruitment of New Neurons in the Postembryonic Optic Tectum
Postembryonic brain development is sensitive to environmental input and sensory experience, but the mechanisms underlying healthy adaptive brain growth are poorly understood. Here, we tested the importance of visual experience on larval zebrafish (Danio rerio) postembryonic development of the optic tectum (OT), a midbrain structure involved in visually guided behavior. We first characterized postembryonic neurogenic growth in OT, in which new neurons are generated along the caudal tectal surface and contribute appositionally to anatomical growth. Restricting visual experience during development by rearing larvae in dim light impaired OT anatomical and neurogenic growth, specifically by reducing the survival of new neurons in the medial periventricular gray zone. Neuronal survival in the OT was reduced only when visual experience was restricted for the first 5 d following new neuron generation, suggesting that tectal neurons exhibit an early sensitive period in which visual experience protects these cells from subsequent neuronal loss. The effect of dim rearing on neuronal survival was mimicked by treatment with an NMDA receptor antagonist early, but not later, in a new neuron’s life. Both dim rearing and antagonist treatment reduced BDNF production in the OT, and supplementing larvae with exogenous BDNF during dim rearing prevented neuronal loss, suggesting that visual experience protects new tectal neurons through neural activity-dependent BDNF expression. Collectively, we present evidence for a sensitive period of neurogenic adaptive growth in the larval zebrafish OT that relies on visual experience-dependent mechanisms.
SIGNIFICANCE STATEMENT Early brain development is shaped by environmental factors via sensory input; however, this form of experience-dependent neuroplasticity is traditionally studied as structural and functional changes within preexisting neurons. Here, we found that restricting visual experience affects development of the larval zebrafish optic tectum, a midbrain structure involved in visually guided behavior, by limiting the survival of newly generated neurons. We found that new tectal neurons exhibit a sensitive period soon after cell birth in which adequate visual experience, likely mediated by neuronal activity driving BDNF production within the tectum, would protect them from subsequent neuronal loss over the following week. Collectively, we present evidence for neurogenic adaptive tectal growth under different environmental lighting conditions.






