Summary: Bilirubin, a bile pigment most commonly associated with jaundice in newborns, appears to have neuroprotective properties. A new study in mice reveals bilirubin may protect the brain against oxidative stress.
Source: Johns Hopkins University
In studies in mice, Johns Hopkins Medicine researchers report they have found that bilirubin, a bile pigment most commonly known for yellowing the skin of people with jaundice, may play an unexpected role in protecting brain cells from damage from oxidative stress.
Bilirubin is commonly measured in lab tests as a marker for liver or blood health, and high levels may indicate disease. However, whether it has a role in healthy people has remained unclear.
The Johns Hopkins Medicine team says its interest in the compound’s function in the brain arose from testing which tissues in the mouse body produced bilirubin. Surprisingly, the researchers found “exceptional levels” of the stuff in mouse brains — five to 10 times higher production than in rodents’ livers.
“Bilirubin is normally considered a waste product, but this level of production takes a lot of metabolic energy, and it seemed bizarre for bilirubin to not have a function,” says Bindu Paul, Ph.D., faculty research instructor at the Johns Hopkins University School of Medicine’s Solomon H. Snyder Department of Neuroscience, and a member of the research team.
The new study, described in a report published July 25 in Cell Chemical Biology, set out to find the purpose for harboring so much bilirubin in the brain. The team noted that past studies proposed that bilirubin might be an important antioxidant. Since the brain is so metabolically active and vulnerable to oxidative damage, the research group considered the possibility that bilirubin might be particularly important to protecting the brain against oxidative stress.
For their experiments, the team used mouse neurons grown in the laboratory that were genetically engineered to not produce bilirubin. As the cells grew, the researchers exposed them to various sources of oxidative stress by introducing reactive molecules to their environment.
When compared with normal mouse brain cells, the researchers found that the genetically modified mouse neurons were far more vulnerable to these stressors — particularly at the hand of a harmful form of oxygen called superoxide.
Chirag Vasavda, an M.D./Ph.D. student in Solomon Snyder’s laboratory and first author on the study, notes that superoxide is an important chemical cell messenger linked to learning, memory and development in the brain.
However, excessive brain cell activity can lead to uncontrolled superoxide levels, which can trigger oxidative stress and initiate a series of harmful reactions that cause damage to the brain. “Our initial experiments hinted to us that bilirubin might play an important role in controlling the levels of superoxide in the brain,” says Vasavda.
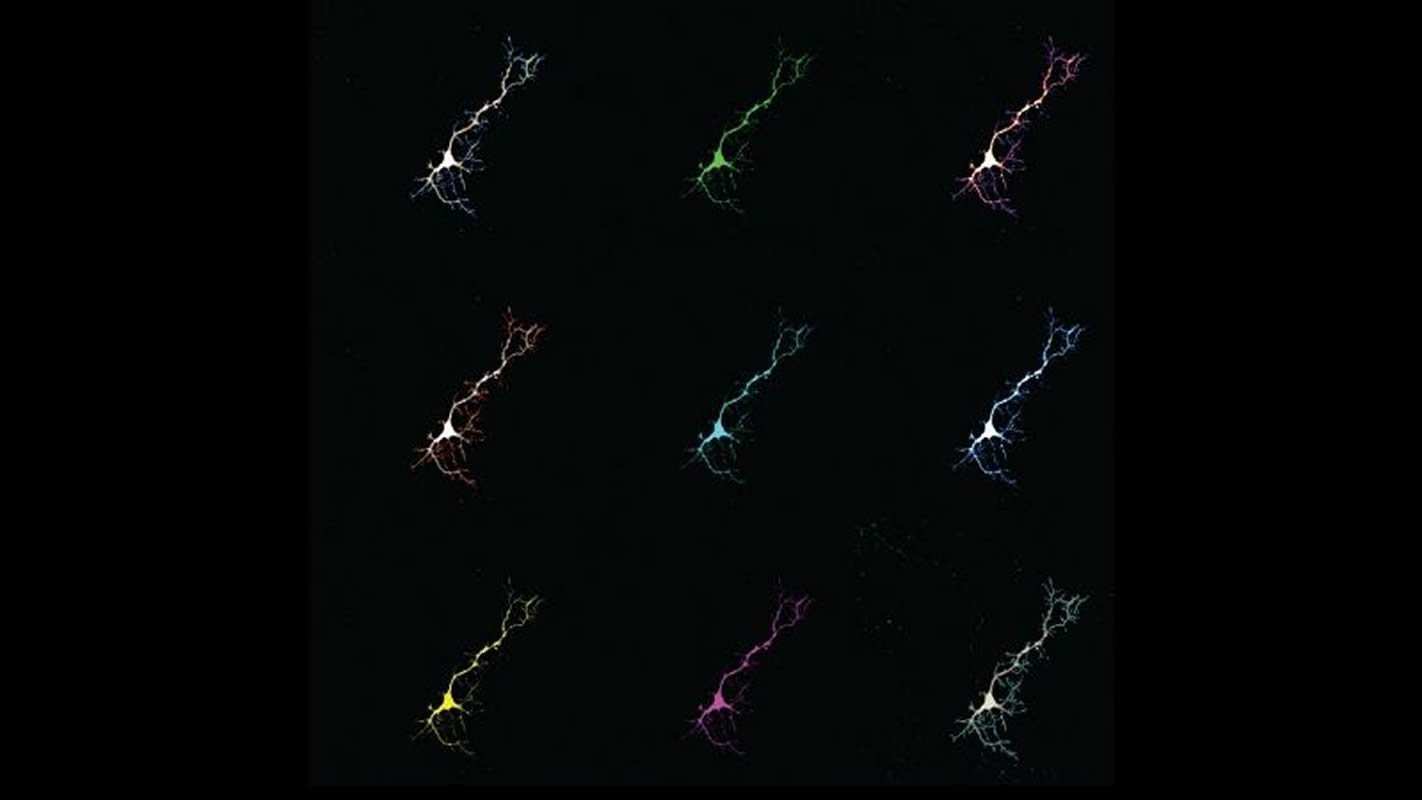
The research team suspected that bilirubin’s ability to regulate superoxide originated in its chemical structure, which allows it to grab on to and neutralize the harmful molecule in a way that other antioxidants, such as glutathione and cysteine, cannot.
To test this, the researchers stimulated excessive brain cell activity in normal brains and brains engineered to lack bilirubin. They found that brains lacking the bilirubin-production gene accumulated excessive superoxide. Then they stimulated brain activity in normal mice and mice lacking bilirubin to test whether removing bilirubin worsens brain damage or cell death.
The researchers found that mice that lacked bilirubin had about two to three times more brain damage as their normal counterparts, suggesting that bilirubin protected normal brains against harmful superoxide reactions.
This discovery, the investigators say, advances scientific understanding of bilirubin’s role in the brain and elsewhere and could lead to novel treatments for neurodegenerative diseases such as Huntington’s and Parkinson’s that are marked by excessive superoxide levels and oxidative stress.
Other researchers involved in this study include Ruchita Kothari, Adarsha Malla, Ming Ji, Cristina Ricco, Risheng Xu, Harry Saavedra, Juan Sbodio, Adele Snowman, Lauren Albacarys, Lynda Hester, Thomas Sedlak and Solomon H. Snyder of the Johns Hopkins University School of Medicine; Robert Tokhunts of Dartmouth College; and Anthony Lin of the Duke University School of Medicine.
Funding: This work was supported by the National Institute of Mental Health (MH18510), the National Institute of General Medical Sciences (T32 GM73009) and the National Institutes of Health Office of the Director (S1D016374, S1OD016374).
The authors declare no competing financial interests.
Source:
Johns Hopkins University
Media Contacts:
Rachel Butch – Johns Hopkins University
Image Source:
The image is credited to Chirag Vasavda.
Original Research: Closed access
“Bilirubin Links Heme Metabolism to Neuroprotection by Scavenging Superoxide”. Chirag Vasavda et al.
Cell Chemical Biology. doi:10.1016/j.chembiol.2019.07.006
Abstract
Bilirubin Links Heme Metabolism to Neuroprotection by Scavenging Superoxide
Highlights
• Eliminating endogenous bilirubin renders cells vulnerable to oxidative stress
• Bilirubin exhibits distinct redox activity among endogenous small molecules
• Bilirubin scavenges O 2 ⋅−
• Neural bilirubin prevents NMDA receptor excitotoxicity by scavenging O 2 ⋅−
Summary
Bilirubin is one of the most frequently measured metabolites in medicine, yet its physiologic roles remain unclear. Bilirubin can act as an antioxidant in vitro, but whether its redox activity is physiologically relevant is unclear because many other antioxidants are far more abundant in vivo. Here, we report that depleting endogenous bilirubin renders mice hypersensitive to oxidative stress. We find that mice lacking bilirubin are particularly vulnerable to superoxide (O 2 ⋅−) over other tested reactive oxidants and electrophiles. Whereas major antioxidants such as glutathione and cysteine exhibit little to no reactivity toward O 2 ⋅−, bilirubin readily scavenges O 2 ⋅−. We find that bilirubin’s redox activity is particularly important in the brain, where it prevents excitotoxicity and neuronal death by scavenging O 2 ⋅− during NMDA neurotransmission. Bilirubin’s unique redox activity toward O 2 ⋅− may underlie a prominent physiologic role despite being significantly less abundant than other endogenous and exogenous antioxidants.






