A brain protein believed to be a key component in the progress of dementia can cause memory loss in healthy brains even before physical signs of degeneration appear according to new University of Sussex research.
The study, published May 29 in the open access Nature Publishing Group journal Scientific Reports, reveals a direct link between the main culprit of Alzheimer’s disease and memory loss.
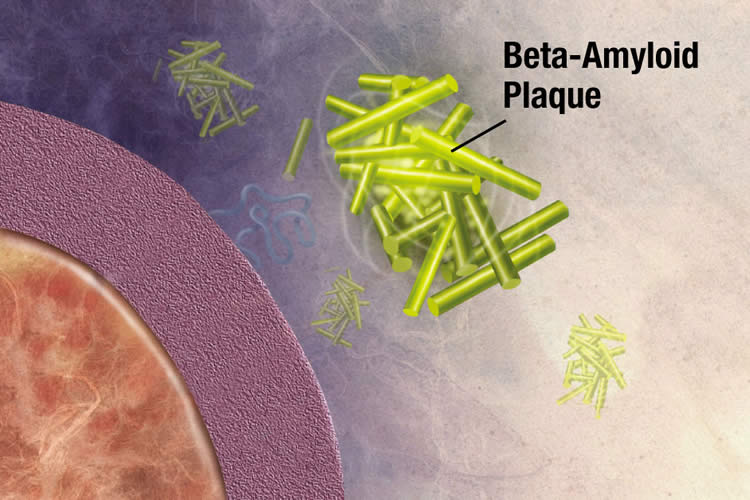
Alzheimer’s disease is characterized by the formation of amyloid plaques in the brain tissue. These amyloid plaques are made up of an insoluble protein, ‘Amyloid-beta’ (Abeta), which forms small structures called ‘oligomers’ that are important in the disease progression.
Although these proteins are known to be involved in Alzheimer’s, little is understood about how they lead to memory loss.
Sussex Neuroscience researchers investigated how Abeta affected healthy brains of pond snails (Lymnaea stagnalis) by observing the effect of administering the protein following a food-reward training task.
The results showed that snails treated with Abeta had significantly impaired memories 24 hours later when tested with the food task, even though their brain tissue showed no sign of damage.
Lead author on the study Lenzie Ford said this demonstrated that Abeta alone is enough to lead to the symptoms of memory loss that are well known in Alzheimer’s disease.
She said, ‘what we observed was that snail brains remained apparently healthy even after the application of the protein. There was no loss of brain tissue, no signs of cell death, no changes in the normal behaviour of the animals, and yet memory was lost.
‘This shows that Alzheimer’s amyloid proteins don’t just affect memory by killing neurons of the brain, they seem to be targeting specific molecular pathways necessary for memories to be preserved.’
Professor George Kemenes, a Sussex neuroscientist who pioneered a thorough understanding of the molecular mechanisms of learning and memory in the pond snail’s nervous system, said, ‘because we understand the memory pathways so well, the simple snail brain has provided the ideal model system to enable us to link the loss of established memory to pure Abeta’.
The work will provide a platform for a more thorough investigation of the mechanisms and effects on memory pathways that lead to this memory loss.
Professor Serpell, a senior author on the study and co-director of the University of Sussex’s Dementia Research Group, said, ‘It is absolutely essential that we understand how Alzheimer’s disease develops in order to find specific targets for therapeutics to combat this disease.’
The data used in the study, “Excess of Rare, Inherited Truncating Mutations in Autism” came from families participating in the Simons Simplex Collection and from the Simons Foundation Autism Research Initiative. The data set including millions of genetic variants, was generated in collaboration with the National Database of Autism Research , and has been made freely available to other autism researchers.
Funding: This study was funded by the Alzheimer’s Research UK, Medical Research Council, Biotechnology and Biological Sciences Research Council, Brighton and Hove City Council, and the American Friends of the University of Sussex.
Source: Jacqui Bealing – University of Sussex
Image Credit: The image is credited to NIA/NIH and is in the public domain
Original Research: Full open access research for “Effects of Aß exposure on long-term associative memory and its neuronal mechanisms in a defined neuronal network” by Lenzie Ford, Michael Crossley, Thomas Williams, Julian R. Thorpe, Louise C. Serpell and György Kemenes in Scientific Reports. Published online May 29 2015 doi:10.1038/srep10614
Abstract
Effects of Aß exposure on long-term associative memory and its neuronal mechanisms in a defined neuronal network
Amyloid beta (Aβ) induced neuronal death has been linked to memory loss, perhaps the most devastating symptom of Alzheimer’s disease (AD). Although Aβ-induced impairment of synaptic or intrinsic plasticity is known to occur before any cell death, the links between these neurophysiological changes and the loss of specific types of behavioral memory are not fully understood. Here we used a behaviorally and physiologically tractable animal model to investigate Aβ-induced memory loss and electrophysiological changes in the absence of neuronal death in a defined network underlying associative memory. We found similar behavioral but different neurophysiological effects for Aβ 25-35 and Aβ 1-42 in the feeding circuitry of the snail Lymnaea stagnalis. Importantly, we also established that both the behavioral and neuronal effects were dependent upon the animals having been classically conditioned prior to treatment, since Aβ application before training caused neither memory impairment nor underlying neuronal changes over a comparable period of time following treatment.
“Effects of Aß exposure on long-term associative memory and its neuronal mechanisms in a defined neuronal network” by Lenzie Ford, Michael Crossley, Thomas Williams, Julian R. Thorpe, Louise C. Serpell and György Kemenes in Scientific Reports. Published online May 29 2015 doi:10.1038/srep10614