Summary: A brain protein genetically lined to Alzheimer’s appears to limit the ability of amyloid beta to trigger synaptic dysfunction, a new study reports.
Source: SBP.
Researchers at Sanford Burnham Prebys Medical Discovery Institute (SBP) have identified a new protective function for a brain protein genetically linked to Alzheimer’s. The findings, published in the Journal of Experimental Medicine, could inform novel treatment strategies.
“We found that a protein called SORLA directly limits the ability of amyloid beta, the toxic protein that causes Alzheimer’s, to trigger the destruction of neuronal connections,” says Huaxi Xu, Ph.D., professor and the Jeanne and Gary Herberger Leadership Chair of SBP’s Neuroscience and Aging Research Center. (SORLA stands for sortilin-related receptor with LDLR class A repeats.) “This is actually the third way that SORLA has been shown to defend against neurodegeneration.”
“It’s becoming increasingly clear that the SORLA gene has a major influence on Alzheimer’s development–more and more Alzheimer’s-associated mutations in the SORLA gene are being discovered,” Xu adds. “Our findings help explain why they are so important.”
SORLA is one of many genes in which mutations are associated with increased risk of Alzheimer’s, which affects 5.5 million people in the U.S. The biggest risk factor is age–as the average life expectancy increases, the number of people with Alzheimer’s is expected to almost triple by 2050.
Alzheimer’s begins when amyloid beta aggregates into small clusters outside neurons. Those clusters, called oligomers, induce toxic signaling that damages the connections between synapses so that neurons can no longer talk to one another. Synapse loss is the reason Alzheimer’s patients develop memory problems.
Xu and his collaborators suspected that SORLA–a trafficking protein that shuttles molecules between cellular compartments–might help protect against amyloid beta induced toxic signaling based on their prior observations. SORLA has already been shown to counteract production of amyloid beta and eliminate it from the space around neurons.
Xu’s team recently reported that SORLA physically interacts with EphA4, one of the receptors through which amyloid beta provokes synaptic dysfunction. (EphA4 exists primarily to control the wiring of neuronal networks as the brain develops and regulate the behavior of synapses in the adult brain.)
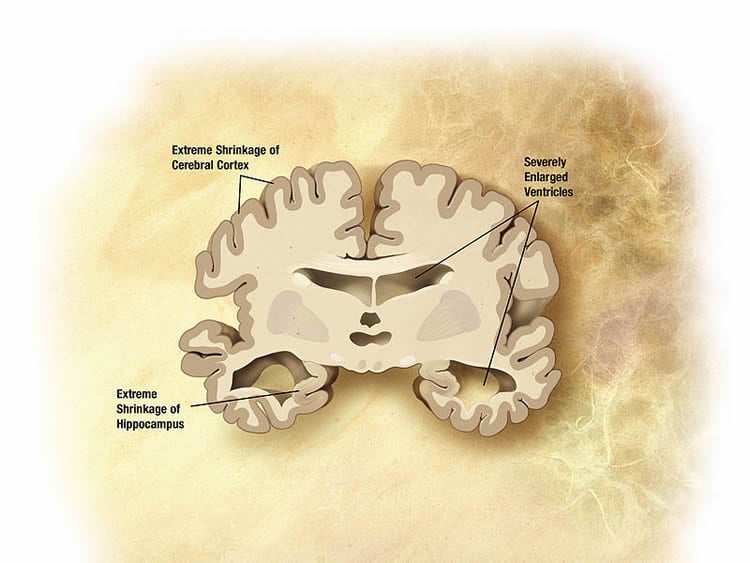
In this study, Xu’s team established that SORLA could mitigate the toxic EphA4 signaling caused by amyloid beta. They also showed that increasing levels of SORLA in mice reduced cognitive impairments caused by amyloid beta.
“These observations suggest that early-stage Alzheimer’s could be treated with drugs that increase levels of SORLA, or that enhance its interaction with EphA4,” comments Xu. “We’re currently searching for drugs that have either of these effects.
“The researchers also found that EphA4 is over-activated in brain tissue from Alzheimer’s patients, and that over-activation correlates with decreased binding to SORLA, demonstrating the relevance of this discovery to human disease.
“Our study also provides support to explore EphA4 inhibitors as Alzheimer’s therapeutics,” Xu notes. “There’s preclinical data from disease models suggesting they have some efficacy.”
“SORLA is becoming a hot topic in Alzheimer’s research. No other protein has yet been found to influence Alzheimer’s pathogenesis in so many ways. And it may do even more–we plan to explore whether it modulates other cell surface amyloid beta receptors such as the cellular prion protein and the NMDA receptor.”
Co-authors of the paper include Timothy Huang (lead author, SBP) Yingjun Zhao (co-lead author, SBP), Lu-lin Jiang (SBP), Xiaoguang Li (SBP), Yan Liu (SBP and Medical College at Xiamen University, China), Yu Sun (SBP), Juan Pina-Crespo (SBP), Bing Zhu (SBP), Elena Pasquale (SBP and UC San Diego) Eliezer Masliah (UC San Diego) Thomas E. Willnow (Max-Delbrueck-Center for Molecular Medicine.)
Funding: Financial support was provided by the National Institutes of Health, the Alzheimer’s Association, the Tanz Family Foundation, the Cure Alzheimer’s Fund, and the Shiley-Marcos Alzheimer’s Disease Research Center.
Source: Susan Gammon – SBP
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is in the public domain.
Original Research: The study will appear in Journal of Experimental Medicine.
[cbtabs][cbtab title=”MLA”]SBP “How SORLA Protects Against Alzheimer’s.” NeuroscienceNews. NeuroscienceNews, 7 November 2017.
<https://neurosciencenews.com/alzheimers-sorla-7894/>.[/cbtab][cbtab title=”APA”]SBP (2017, November 7). How SORLA Protects Against Alzheimer’s. NeuroscienceNews. Retrieved November 7, 2017 from https://neurosciencenews.com/alzheimers-sorla-7894/[/cbtab][cbtab title=”Chicago”]SBP “How SORLA Protects Against Alzheimer’s.” https://neurosciencenews.com/alzheimers-sorla-7894/ (accessed November 7, 2017).[/cbtab][/cbtabs]






