Summary: Changes in the brain’s serotonin system may occur years before Parkinson’s disease symptoms begin. Serotonin alterations could be used as a biomarker for Parkinson’s disease.
Source: Kings College London
Researchers from King’s College London have uncovered the earliest signs of Parkinson’s disease in the brain, many years before patients show any symptoms. The results, published in The Lancet Neurology, challenge the traditional view of the disease and could potentially lead to screening tools for identifying people at greatest risk.
Parkinson’s disease is the second most common neurodegenerative disorder, after Alzheimer’s disease. The disease is characterised by movement and cognitive problems but is known to become established in the brain a long time before patients are diagnosed. Studying the crucial early stages of the disease, when treatment could potentially slow its progress, is a huge challenge.
The new study, funded by the Lily Safra Foundation, provides the first evidence of a central role for the brain chemical serotonin in the very earliest stages of Parkinson’s. The results suggest changes to the serotonin system could act as a key early warning signal for the disease.
Chief investigator Professor Marios Politis, Lily Safra Professor of Neurology & Neuroimaging at the Institute of Psychiatry, Psychology & Neuroscience (IoPPN), says: ‘Parkinson’s disease has traditionally been thought of as occurring due to damage in the dopamine system, but we show that changes to the serotonin system come first, occurring many years before patients begin to show symptoms. Our results suggest that early detection of changes in the serotonin system could open doors to the development of new therapies to slow, and ultimately prevent, the progression of Parkinson’s disease.’
People with Parkinson’s disease have build-ups of the protein α-synuclein in the brain. While there is no clear cause for most people, a minority of cases are caused by genetic mutations. People with mutations in the α-synuclein (SNCA) gene are extremely rare but are almost certain to develop Parkinson’s disease during their lifetime, making them ideal for studying the train of biological events that leads to Parkinson’s disease.
The SNCA genetic mutation originates in villages in the northern Peloponnese in Greece and can also be found in people who migrated to nearby regions in Italy. Over two years, the researchers identified 14 people with the SNCA gene mutation from Greece and Italy and flew them to London for brain imaging and clinical assessments. Half of the participants had not begun to show any symptoms of Parkinson’s.
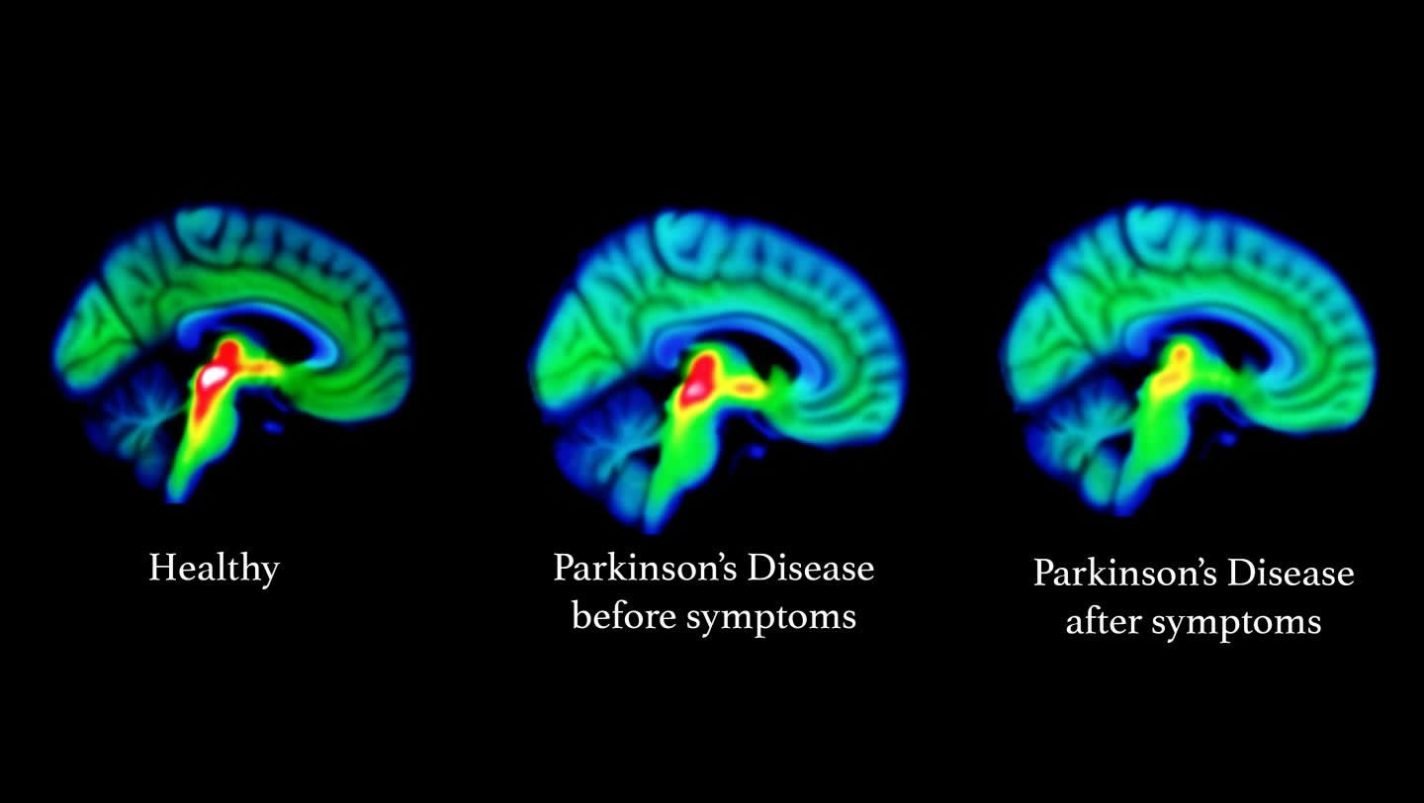
Data from the 14 people with SNCA gene mutations were compared with 65 patients with non-genetic Parkinson’s disease and 25 healthy volunteers. The researchers found that the serotonin system starts to malfunction in people with Parkinson’s well before symptoms affecting movement occur, and before the first changes in the dopamine system.
First author Heather Wilson, from the IoPPN, says: ‘We found that serotonin function was an excellent marker for how advanced Parkinson’s disease has become. Crucially, we found detectable changes to the serotonin system among patients who were not yet diagnosed. Therefore, brain imaging of the serotonin system could become a valuable tool to detect individuals at risk for Parkinson’s disease, monitor their progression and help with the development of new treatments.’
Brain imaging was carried out using PET scans which are expensive and difficult to carry out. The researchers say further work is required to develop the scanning techniques in order to make them more affordable and straightforward for use as screening tools.
Source:
Kings College London
Media Contacts:
Robin Bisson – Kings College London
Image Source:
The images are credited to Neurodegeneration Imaging Group, King’s College London.
Original Research: Closed access
“Serotonergic pathology and disease burden in the premotor and motor phase of A53T α-synuclein parkinsonism: a cross-sectional study”. Marios Politis et al.
The Lancet Neurology. doi:10.1016/S1474-4422(19)30140-1
Abstract
Serotonergic pathology and disease burden in the premotor and motor phase of A53T α-synuclein parkinsonism: a cross-sectional study
Background
Because of the highly penetrant gene mutation and clinical features consistent with idiopathic Parkinson’s disease, carriers of the autosomal dominant Ala53Thr (A53T; 209G→A) point mutation in the α-synuclein ( SNCA) gene are an ideal population to study the premotor phase and evolution of Parkinson’s pathology. Given the known neurochemical changes in the serotonergic system and their association with symptoms of Parkinson’s disease, we hypothesised that carriers of the A53T SNCA mutation might show abnormalities in the serotonergic neurotransmitter system before the diagnosis of Parkinson’s disease, and that this pathology might be associated with measures of Parkinson’s burden.
Methods
In this cross-sectional study, we recruited carriers of the A53T SNCA mutation from specialist Movement Disorders clinics in Athens, Greece, and Salerno, Italy, and a cohort of healthy controls with no personal or family history of neurological or psychiatric disorders from London, UK (recruited via public advertisement) who were age matched to the A53T SNCA carriers. We also recruited one cohort of patients with idiopathic Parkinson’s disease (cohort 1) from Movement Disorders clinics in London, UK, and retrieved data on a second cohort of such patients (cohort 2; n=40) who had been scanned with a different scanner. 7-day continuous recording of motor function was used to determine the Parkinson’s disease status of the A53T carriers. To assess whether serotonergic abnormalities were present, we used [ 11C]DASB PET non-displaceable binding to quantify serotonin transporter density. We constructed brain topographic maps reflecting Braak stages 1–6 and used these as seed maps to calculate [ 11C]DASB non-displaceable binding potential in our cohort of A53T SNCA carriers. Additionally, all participants underwent a battery of clinical assessments to determine motor and non-motor symptoms and cognitive status, and [ 123I]FP-CIT single-photon emission CT (SPECT) to assess striatal dopamine transporter binding and MRI for volumetric analyses to assess whether pathology is associated with measures of Parkinson’s disease burden.
Findings
Between Sept 1, 2016, and Sept 30, 2018, we recruited 14 A53T SNCA carriers, 25 healthy controls, and 25 patients with idiopathic Parkinson’s disease. Seven (50%) of 14 A53T SCNA carriers were confirmed to have motor symptoms and confirmed to have Parkinson’s disease, and the absence of motor symptoms was confirmed in seven (50%) A53T SCNA carriers (ie, premotor), in whom [ 123I]FP-CIT SPECT confirmed the absence of striatal dopaminergic deficits. Compared with healthy controls, premotor A53T SNCA carriers showed loss of [ 11C]DASB non-displaceable binding potential in the ventral (p<0·0001) and dorsal (p=0·0002) raphe nuclei, caudate (p=0·00015), putamen (p=0·036), thalamus (p=0·00074), hypothalamus (p<0·0001), amygdala (p=0·0041), and brainstem (p=0·046); and in A53T SNCA carriers with Parkinson's disease this loss was extended to the hippocampus (p=0·0051), anterior (p=0·022) and posterior cingulate (p=0·036), insula (p=0·0051), frontal (p=0·0016), parietal (p=0·019), temporal (p<0·0001), and occipital (p=0·0053) cortices. A53T SNCA carriers with Parkinson's disease showed a loss of striatal [ 123I]FP-CIT-specific binding ratio compared with healthy controls (p<0·0001). Premotor A53T SNCA carriers had loss of [ 11C]DASB non-displaceable binding potential in brain areas corresponding to Braak stages 1–3, whereas [ 11C]DASB non-displaceable binding potential was largely preserved in areas corresponding to Braak stages 4–6. Except for one participant who was diagnosed with Parkinson's disease in the past year, all A53T SNCA carriers with Parkinson's disease had decreases in [ 11C]DASB non-displaceable binding potential in brain areas corresponding to Braak stages 1–6. Decreases in [ 11C]DASB non-displaceable binding potential in the brainstem were associated with increased Movement Disorder Score-Unified Parkinson's Disease Rating Scale total scores in all A53T SNCA carriers ( r −0·66, 95% CI −0·88 to −0·20; p=0·0099), idiopathic Parkinson's disease cohort 1 ( r −0·66, −0·84 to −0·36; p=0·00031), and idiopathic Parkinson's disease cohort 2 ( r −0·71, −0·84 to −0·52; p<0·0001).
Interpretation
The presence of serotonergic pathology in premotor A53T SNCA carriers preceded development of dopaminergic pathology and motor symptoms and was associated with disease burden, highlighting the potential early role of serotonergic pathology in the progression of Parkinson’s disease. Our findings provide evidence that molecular imaging of serotonin transporters could be used to visualise premotor pathology of Parkinson’s disease in vivo. Future work might establish whether serotonin transporter imaging is suitable as an adjunctive tool for screening and monitoring progression for individuals at risk or patients with Parkinson’s disease to complement dopaminergic imaging, or as a marker of Parkinson’s burden in clinical trials.
Funding
Lily Safra Hope Foundation and National Institute for Health Research (NIHR) Biomedical Research Centre at King’s College London.







