Summary: A new study reveals sleep deprivation can lead to an immediate increase in amyloid beta.
Source: NIH/NIAAA.
Losing just one night of sleep led to an immediate increase in beta-amyloid, a protein in the brain associated with Alzheimer’s disease, according to a small, new study by researchers at the National Institutes of Health. In Alzheimer’s disease, beta-amyloid proteins clump together to form amyloid plaques, a hallmark of the disease.
While acute sleep deprivation is known to elevate brain beta-amyloid levels in mice, less is known about the impact of sleep deprivation on beta-amyloid accumulation in the human brain. The study is among the first to demonstrate that sleep may play an important role in human beta-amyloid clearance.
“This research provides new insight about the potentially harmful effects of a lack of sleep on the brain and has implications for better characterizing the pathology of Alzheimer’s disease,” said George F. Koob, Ph.D., director of the National Institute on Alcohol Abuse and Alcoholism (NIAAA), part of the National Institutes of Health, which funded the study.
Beta-amyloid is a metabolic waste product present in the fluid between brain cells. In Alzheimer’s disease, beta-amyloid clumps together to form amyloid plaques, negatively impacting communication between neurons.
Led by Drs. Ehsan Shokri-Kojori and Nora D. Volkow of the NIAAA Laboratory of Neuroimaging, the study is now online in the Proceedings of the National Academy of Sciences. Dr. Volkow is also the director of the National Institute on Drug Abuse at NIH.
To understand the possible link between beta-amyloid accumulation and sleep, the researchers used positron emission tomography (PET) to scan the brains of 20 healthy subjects, ranging in age from 22 to 72, after a night of rested sleep and after sleep deprivation (being awake for about 31 hours). They found beta-amyloid increases of about 5 percent after losing a night of sleep in brain regions including the thalamus and hippocampus, regions especially vulnerable to damage in the early stages of Alzheimer’s disease.
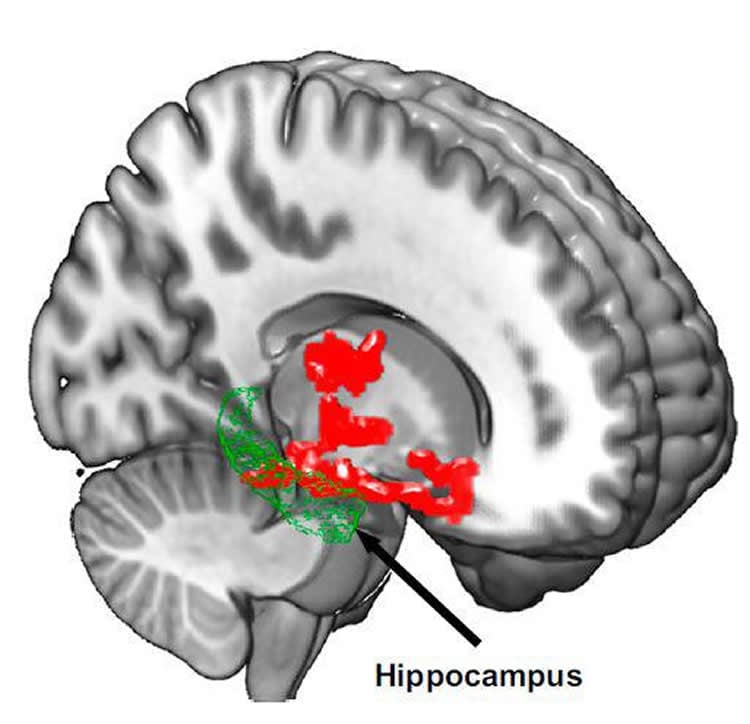
In Alzheimer’s disease, beta-amyloid is estimated to increase about 43 percent in affected individuals relative to healthy older adults. It is unknown whether the increase in beta-amyloid in the study participants would subside after a night of rest.
The researchers also found that study participants with larger increases in beta-amyloid reported worse mood after sleep deprivation.
“Even though our sample was small, this study demonstrated the negative effect of sleep deprivation on beta-amyloid burden in the human brain. Future studies are needed to assess the generalizability to a larger and more diverse population,” said Dr. Shokri-Kojori.
It is also important to note that the link between sleep disorders and Alzheimer’s risk is considered by many scientists to be “bidirectional,” since elevated beta-amyloid may also lead to sleep disturbances.
Funding: NIH/National Institute on Alcohol Abuse and Alcoholism funded this study.
Source: NIH/NIAAA
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to PNAS.
Original Research: Open access research for “β-Amyloid accumulation in the human brain after one night of sleep deprivation” by Ehsan Shokri-Kojori, Gene-Jack Wang, Corinde E. Wiers, Sukru B. Demiral, Min Guo, Sung Won Kim, Elsa Lindgren, Veronica Ramirez, Amna Zehra, Clara Freeman, Gregg Miller, Peter Manza, Tansha Srivastava, Susan De Santi, Dardo Tomasi, Helene Benveniste and Nora D. Volkow in PNAS. Published April 9 2018,
doi:10.1073/pnas.1721694115
[cbtabs][cbtab title=”MLA”]NIH/NIAAA “Lack of Sleep May Be Linked to Risk Factor for Alzheimer’s.” NeuroscienceNews. NeuroscienceNews, 13 April 2018.
<https://neurosciencenews.com/sleep-alzheimers-risk-8782/>.[/cbtab][cbtab title=”APA”]NIH/NIAAA (2018, April 13). Lack of Sleep May Be Linked to Risk Factor for Alzheimer’s. NeuroscienceNews. Retrieved April 13, 2018 from https://neurosciencenews.com/sleep-alzheimers-risk-8782/[/cbtab][cbtab title=”Chicago”]NIH/NIAAA “Lack of Sleep May Be Linked to Risk Factor for Alzheimer’s.” https://neurosciencenews.com/sleep-alzheimers-risk-8782/ (accessed April 13, 2018).[/cbtab][/cbtabs]
Abstract
β-Amyloid accumulation in the human brain after one night of sleep deprivation
The effects of acute sleep deprivation on β-amyloid (Aβ) clearance in the human brain have not been documented. Here we used PET and 18F-florbetaben to measure brain Aβ burden (ABB) in 20 healthy controls tested after a night of rested sleep (baseline) and after a night of sleep deprivation. We show that one night of sleep deprivation, relative to baseline, resulted in a significant increase in Aβ burden in the right hippocampus and thalamus. These increases were associated with mood worsening following sleep deprivation, but were not related to the genetic risk (APOE genotype) for Alzheimer’s disease. Additionally, baseline ABB in a range of subcortical regions and the precuneus was inversely associated with reported night sleep hours. APOE genotyping was also linked to subcortical ABB, suggesting that different Alzheimer’s disease risk factors might independently affect ABB in nearby brain regions. In summary, our findings show adverse effects of one-night sleep deprivation on brain ABB and expand on prior findings of higher Aβ accumulation with chronic less sleep.






