In rhesus monkey families — just as in their human cousins — anxious parents are more likely to have anxious offspring.
A new study in an extended family of monkeys provides important insights into how the risk of developing anxiety and depression is passed from parents to children.
The study from the Department of Psychiatry and the HealthEmotions Research Institute at the University of Wisconsin-Madison shows how an over-active brain circuit involving three brain areas inherited from generation to generation may set the stage for developing anxiety and depressive disorders.
The study is being published this week in the Proceedings of the National Academy of Sciences (PNAS). It shows that elevated activity in this prefrontal-limbic-midbrain circuit is likely involved in mediating the in-born risk for extreme anxiety, anxious temperament that can be observed in early childhood.
“Over-activity of these three brain regions are inherited brain alterations that are directly linked to the later life risk to develop anxiety and depression,” says senior author Ned Kalin, chair of psychiatry at the UW School of Medicine and Public Health. “This is a big step in understanding the neural underpinnings of inherited anxiety and begins to give us more selective targets for treatment.”
Previous research by Kalin’s group has shown that anxious temperament is inherited, and explained the brain circuits involved. About half of children who show extreme anxiety go on to develop stress-related psychiatric disorders later in life.
Monkeys, like humans, can be temperamentally anxious and pass their anxiety-related genes on to the next generation.
By studying nearly 600 young rhesus monkeys from a large multi-generational family, Andrew Fox, Kalin, and colleagues found that about 35 percent of variation in anxiety-like tendencies is explained by family history.
To understand which brain regions are responsible for passing anxiety from generation to generation, the authors measured anxiety-related behavior with high-resolution functional and structural brain imaging. They exposed the young monkeys to a mildly threatening situation that a child would also encounter, exposure to a stranger who does not make eye contact with the monkey. During this encounter, they used imaging methods commonly used in humans (positron emission tomography, PET) to identify brain regions in which increased metabolism predicted each individual’s level of anxiety.
By closely examining how individual differences in brain function and anxiety-related behavior fall through the family tree, the authors identified brain systems responsible for the parent-to-child transmission of anxiety-related behavior. Using this “genetic correlation” approach, the authors found the neural circuit where metabolism and an early-life anxious temperament are likely to share the same genetic basis.
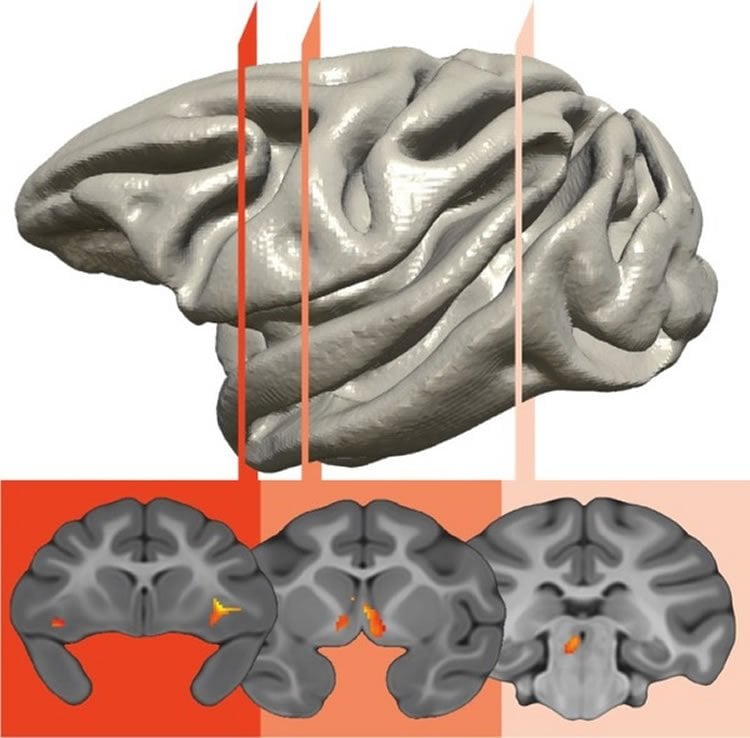
Interestingly, the brain circuit that was genetically correlated with individual differences in early-life anxiety involved three survival-related brain regions. These regions were located in the brain stem, the most primitive part of the brain; the amygdala, the limbic brain fear center; and the prefrontal cortex, which is responsible for higher-level reasoning and is fully developed only in humans and their primate cousins.
“Basically, we think that to a certain extent, anxiety can provide an evolutionary advantage because it helps an individual recognize and avoid danger, but when the circuits are over-active, it becomes a problem and can result in anxiety and depressive disorders,” Kalin explains.
Surprisingly, these studies found that it was the function of these brain structures — and not their size — that was responsible for the genetic transfer of an anxious temperament. Although the search for the genetic underpinnings of anxiety have thus far been elusive, this research helps explain how genes might affect brain function and lead to extreme childhood anxiety, which greatly increases the risk to develop anxiety and depressive disorders.
“Now that we know where to look, we can develop a better understanding of the molecular alterations that give rise to anxiety-related brain function,” Kalin says. “Our genes shape our brains to help make us who we are.”
Funding: The research was funded by the National Institute of Mental Health (NIMH).
Source: University of Wisconsin-Madison
Image Credit: Image is credited to Kalin Lab
Original Research: Abstract for “Intergenerational Neural Mediators of Early-Life Anxious Temperament” by Andrew S. Fox, Jonathan A. Oler, Alexander J. Shackman, Steven E. Shelton, Muthuswamy Raveendran, D. Reese McKay, Alexander K. Converse, Andrew Alexander, Richard J. Davidson, John Blangero, Jeffrey Rogers, and Ned H. Kalin in PNAS. Published online July 6 2015 doi:10.1073/pnas.1508593112
Abstract
Intergenerational Neural Mediators of Early-Life Anxious Temperament
Understanding the heritability of neural systems linked to psychopathology is not sufficient to implicate them as intergenerational neural mediators. By closely examining how individual differences in neural phenotypes and psychopathology cosegregate as they fall through the family tree, we can identify the brain systems that underlie the parent-to-child transmission of psychopathology. Although research has identified genes and neural circuits that contribute to the risk of developing anxiety and depression, the specific neural systems that mediate the inborn risk for these debilitating disorders remain unknown. In a sample of 592 young rhesus monkeys that are part of an extended multigenerational pedigree, we demonstrate that metabolism within a tripartite prefrontal-limbic-midbrain circuit mediates some of the inborn risk for developing anxiety and depression. Importantly, although brain volume is highly heritable early in life, it is brain metabolism—not brain structure—that is the critical intermediary between genetics and the childhood risk to develop stress-related psychopathology.
“Intergenerational Neural Mediators of Early-Life Anxious Temperament” by Andrew S. Fox, Jonathan A. Oler, Alexander J. Shackman, Steven E. Shelton, Muthuswamy Raveendran, D. Reese McKay, Alexander K. Converse, Andrew Alexander, Richard J. Davidson, John Blangero, Jeffrey Rogers, and Ned H. Kalin in PNAS. Published online July 6 2015 doi:10.1073/pnas.1508593112







