Summary: By transplanting living brain cells from an organoid into newborn rats, researchers create a new method of studying neurodevelopment and show neurons from one species can effectively form connections with another.
Source: Neuroscience News
Transplanting a cluster of living human brain cells from an organoid into the brains of newborn rats, researchers have created a new method to study a range of neurodevelopmental disorders.
Additionally, the new model demonstrates neurons from one species can effectively form neural connections with another species.
Brain organoids are created from human pluripotent stem cells which are grown in a lab to allow for the study of functional brain development. This technique has gained popularity in the study of neurodevelopmental disorders, such as autism and schizophrenia.
However, there are limitations in using brain organoids, namely that the neurons grown in a dish do not grow as large or make as many connections as those found in the human brain.
In order to continue studying brain development from the organoids, researchers were tasked with developing a new method that would allow the cells to develop to a more mature and functional level that mimicked development in the human brain. They turned their attention to newborn rats.
In a new study, published today in Nature, researchers transplanted human-derived brain stem cells into the cerebral cortex of over 80 rat models. Over the course of eight months, the neurons grew to take up one-third of the hemisphere of the rat brains. The researchers reported the neurons grew to be at least six times larger than they generally grow to while in a petri dish. The neurons had greater connectivity than organoid models, and more closely mimicked cells within a human brain compared to traditional organoid neurons.
However, there were some key differences in the transplanted cells compared to naturally developing human neurons. The stem cells did not grow into all types of brain cells found in the human brain and they did not develop layers as cells in the cerebral cortex.
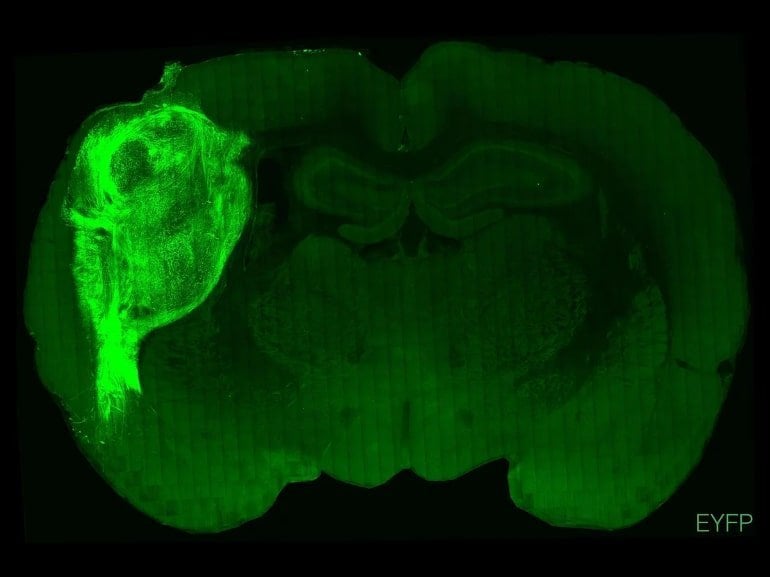
In further experiments, researchers found significant differences between rats that received stem cell transplants and those that did not. When the human-derived neurons were exposed to blue light, the rats could distinguish between red and blue lights, while the natural rats could not. Also, when researchers touched the whiskers near the transplantation site, the human neurons became stimulated.
In testing for neurodevelopmental disorders, researchers utilized organoid stem cell transplantation to create models of Timothy syndrome, a genetic disorder that causes developmental delays. In organoid models, the cells looked almost identical to normal brain cells. In the rat models, the cells displayed abnormal branching and were shorter than unaffected brain cells.
While the findings present a significant breakthrough for the study of developmental disorders, some point to neuroethical issues of cross-species transplantation. Many have questioned whether organoids could reach “human-like consciousness”. This question, researchers say, is a question scientists and bioethicists will have to answer together.
About this stem cell transplantation and neuroscience research news
Author: Press Office
Source: Neuroscience News
Contact: Press Office – Neuroscience News
Image: The image is credited to Pasca lab / Stanford Medicine
Original Research: Open access.
“Maturation and circuit integration of transplanted human cortical organoids” by Sergiu P. Pașca et al. Nature
Abstract
Maturation and circuit integration of transplanted human cortical organoids
Self-organizing neural organoids represent a promising in vitro platform with which to model human development and disease. However, organoids lack the connectivity that exists in vivo, which limits maturation and makes integration with other circuits that control behaviour impossible.
Here we show that human stem cell-derived cortical organoids transplanted into the somatosensory cortex of newborn athymic rats develop mature cell types that integrate into sensory and motivation-related circuits.
MRI reveals post-transplantation organoid growth across multiple stem cell lines and animals, whereas single-nucleus profiling shows progression of corticogenesis and the emergence of activity-dependent transcriptional programs.
Indeed, transplanted cortical neurons display more complex morphological, synaptic and intrinsic membrane properties than their in vitro counterparts, which enables the discovery of defects in neurons derived from individuals with Timothy syndrome.
Anatomical and functional tracings show that transplanted organoids receive thalamocortical and corticocortical inputs, and in vivo recordings of neural activity demonstrate that these inputs can produce sensory responses in human cells.
Finally, cortical organoids extend axons throughout the rat brain and their optogenetic activation can drive reward-seeking behaviour. Thus, transplanted human cortical neurons mature and engage host circuits that control behaviour.
We anticipate that this approach will be useful for detecting circuit-level phenotypes in patient-derived cells that cannot otherwise be uncovered.






