Summary: A new study reports cannabis can prune synapses and destroy some neural circuits.
Source: Osaka University.
A group of researchers led by Associate Professor KIMURA Fumitaka at the Department of Molecular Neuroscience, Graduate School of Medicine, Osaka University, clarified that multiple mechanisms were involved in the formation of neuronal circuits in the cerebral cortex. This group also clarified that a substance similar to cannabinoid, an active substance of cannabis, played an important role in the formation of neuronal circuits, and that the intake of cannabis pruned even necessary synapses, destroying neuronal circuits, a world first.
It was known that neural activities by presynaptic and postsynaptic cells greatly affected the formation of neural circuits, but the kinds of neural activities involved were not clear. Neutral activities in thalamocortical projections to the cortex are complicated, proceeding in two steps. As the cerebral cortex grows, unnecessary projections are pruned, eventually forming correct projections. However, this mechanism was unclear.
This group had discovered that other areas of the cerebral cortex had a rule called spike timing-dependent plasticity (STDP), that is, the synaptic strength was determined in order of firing of presynaptic and postsynaptic cells, and that this rule suddenly changed in one process during circuit formation. For this reason, this group focused on the possibility that the rule would suddenly change at thalamocortical synapses as they performed their research.
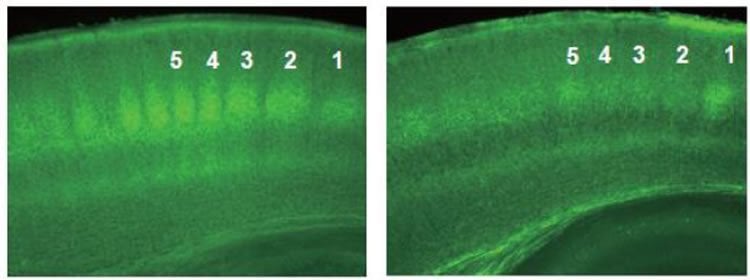
As a result, when thalamocortical projections were formed, first, synaptic connections were strengthened due to activities in presynaptic and postsynaptic cells, and projections extended over wide areas, but following that, the rule changed so that the synchronized activities weakened the synaptic connections, and extra projections were pruned. In addition, it was found that at the time of weakening of synaptic connections, cannabinoid was released from neural cells via synchronized activities, thereby retracting unnecessary neuron projections. Research showed that neuron projections retracted when cannabinoid was externally taken as well. Furthermore, it was also found that projections remained disorderly in cannabinoid receptors (CB1Rs) knock-out mice.
This group’s achievement clarified scientific grounds that cannabis and intoxicating drugs had adverse effects on the brain. By disabling the functions of cannabinoid, it may be possible to suppress the breakdown of neural circuits. It is expected that this group’s achievement will be used for recovering functions impaired by brain damage and dementia.
Funding: NIH//National Institute on Deafness and Other Communication Disorders funded study.
Source: Osaka University
Image Source: This NeuroscienceNews.com image is credited to the researchers.
Original Research: Abstract for “Developmental Switch in Spike Timing-Dependent Plasticity and Cannabinoid-Dependent Reorganization of the Thalamocortical Projection in the Barrel Cortex” by Chiaki Itami, Jui-Yen Huang, Miwako Yamasaki, Masahiko Watanabe, Hui-Chen Lu, and Fumitaka Kimura in Journal of Neuroscience. Published online June 29 2016 doi:10.1523/JNEUROSCI.4280-15.2016
[cbtabs][cbtab title=”MLA”]Osaka University. “How Cannabis Influences Formation of Neural Circuits.” NeuroscienceNews. NeuroscienceNews, 12 September 2016.
<https://neurosciencenews.com/neural-circuits-cannabis-5020/>.[/cbtab][cbtab title=”APA”]Osaka University. (2016, September 12). How Cannabis Influences Formation of Neural Circuits. NeuroscienceNews. Retrieved September 12, 2016 from https://neurosciencenews.com/neural-circuits-cannabis-5020/[/cbtab][cbtab title=”Chicago”]Osaka University. “How Cannabis Influences Formation of Neural Circuits.” https://neurosciencenews.com/neural-circuits-cannabis-5020/ (accessed September 12, 2016).[/cbtab][/cbtabs]
Abstract
Developmental Switch in Spike Timing-Dependent Plasticity and Cannabinoid-Dependent Reorganization of the Thalamocortical Projection in the Barrel Cortex
The formation and refinement of thalamocortical axons (TCAs) is an activity-dependent process (Katz and Shatz, 1996), but its mechanism and nature of activity are elusive. We studied the role of spike timing-dependent plasticity (STDP) in TCA formation and refinement in mice. At birth (postnatal day 0, P0), TCAs invade the cortical plate, from which layers 4 (L4) and L2/3 differentiate at P3-P4. A portion of TCAs transiently reach toward the pia surface around P2-P4 (Senft and Woolsey, 1991; Rebsam et al., 2002) but are eventually confined below the border between L2/3 and L4. We previously showed that L4-L2/3 synapses exhibit STDP with only potentiation (timing-dependent long-term potentiation [t-LTP]) during synapse formation, then switch to a Hebbian form of STDP. Here we show that TCA-cortical plate synapses exhibit robust t-LTP in neonates, whose magnitude decreased gradually after P4-P5. After L2/3 is differentiated, TCA-L2/3 gradually switched to STDP with only depression (t-LTD) after P7-P8, whereas TCA-L4 lost STDP. t-LTP was dependent on NMDA receptor and PKA, whereas t-LTD was mediated by Type 1 cannabinoid receptors (CB1Rs) probably located at TCA terminals, revealed by global and cortical excitatory cell-specific knock-out of CB1R. Moreover, we found that administration of CB1R agonists, including Δ9-tetrahydrocannabinol, caused substantial retraction of TCAs. Consistent with this, individual thalamocortical axons exuberantly innervated L2/3 at P12 in CB1R knock-outs, indicating that endogenous cannabinoid signaling shapes TCA projection. These results suggest that the developmental switch in STDP and associated appearance of CB1R play important roles in the formation and refinement of TCAs.
SIGNIFICANCE STATEMENT It has been shown that neural activity is required for initial synapse formation of thalamocortical axons with cortical cells, but precisely what sort of activities in presynaptic and postsynaptic cells are required is not yet clear. In addition, how activity is further translated into structural changes is unclear. We show here that the period during which spike timing-dependent long-term potentiation and depression (t-LTP, t-LTD) can be induced closely matches the time course of synapse formation and retraction, respectively, at the thalamocortical synapse. Moreover, administration of cannabinoid agonists, which mimic t-LTD, caused TCA retraction, suggesting that cannabinoids translate physiological changes into morphological consequences.
“Developmental Switch in Spike Timing-Dependent Plasticity and Cannabinoid-Dependent Reorganization of the Thalamocortical Projection in the Barrel Cortex” by Chiaki Itami, Jui-Yen Huang, Miwako Yamasaki, Masahiko Watanabe, Hui-Chen Lu, and Fumitaka Kimura in Journal of Neuroscience. Published online June 29 2016 doi:10.1523/JNEUROSCI.4280-15.2016






