Summary: Researchers report myelin continues to form and restructure in the adult brain.
Source: Yale.
Myelin acts as insulation for millions of brain cells, allowing for swift and efficient transmission of signals across brain regions. Despite its crucial role, little is known about how stable this structure is in the adult brain and what impact aging has on its maintenance.
Yale neurologists Robert Hill, Alice Li, and Jaime Grutzendler devised techniques to track and precisely image myelin throughout the lifetime of the mouse. They discovered that myelin continues to form and restructure in the adult brain — indicating the potential for lifelong change. They also learned that during aging, myelin begins to deteriorate and myelin debris accumulate over time.
“Myelin is not static in the adult brain and may play an underappreciated role in brain plasticity, a role that is likely to be disrupted as we age,” Hill said.
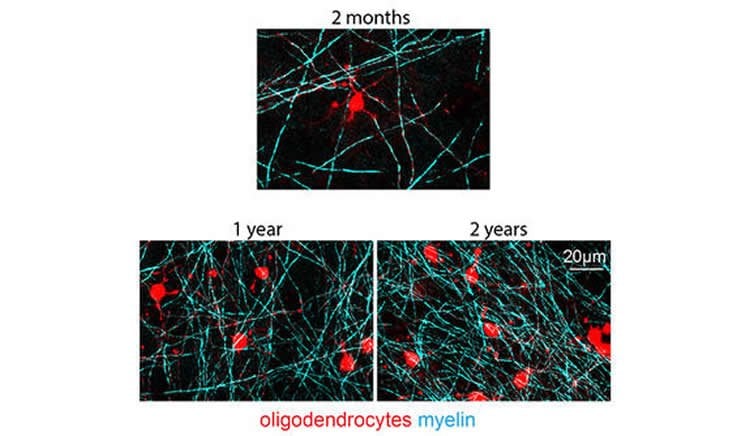
Source: Bill Hathaway – Yale
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to the researchers.
Original Research: Abstract in Nature Neuroscience.
doi:10.1038/s41593-018-0120-6
[cbtabs][cbtab title=”MLA”]Yale “Myelin Continues to Grow Throughout Life.” NeuroscienceNews. NeuroscienceNews, 20 March 2018.
<https://neurosciencenews.com/myelin-regrowth-8679/>.[/cbtab][cbtab title=”APA”]Yale (2018, March 20). Myelin Continues to Grow Throughout Life. NeuroscienceNews. Retrieved March 20, 2018 from https://neurosciencenews.com/myelin-regrowth-8679/[/cbtab][cbtab title=”Chicago”]Yale “Myelin Continues to Grow Throughout Life.” https://neurosciencenews.com/myelin-regrowth-8679/ (accessed March 20, 2018).[/cbtab][/cbtabs]
Abstract
Lifelong cortical myelin plasticity and age-related degeneration in the live mammalian brain
Axonal myelin increases neural processing speed and efficiency. It is unknown whether patterns of myelin distribution are fixed or whether myelinating oligodendrocytes are continually generated in adulthood and maintain the capacity for structural remodeling. Using high-resolution, intravital label-free and fluorescence optical imaging in mouse cortex, we demonstrate lifelong oligodendrocyte generation occurring in parallel with structural plasticity of individual myelin internodes. Continuous internode formation occurred on both partially myelinated and unmyelinated axons, and the total myelin coverage along individual axons progressed up to two years of age. After peak myelination, gradual oligodendrocyte death and myelin degeneration in aging were associated with pronounced internode loss and myelin debris accumulation within microglia. Thus, cortical myelin remodeling is protracted throughout life, potentially playing critical roles in neuronal network homeostasis. The gradual loss of internodes and myelin degeneration in aging could contribute significantly to brain pathogenesis.