Summary: Researchers report helping the immune system clear away old senescent cells in aging mice helped to restore youthful characteristics. The findings could have implications for reversing the effects of aging in humans.
Source: Weizmann Institute of Science.
If only we could keep our bodies young, healthy and energetic, even as we attain the wisdom of our years. New research at the Weizmann Institute of Science suggests this dream could be at least partly obtainable in the future. The results of this research, led by Prof. Valery Krizhanovsky and Dr. Yossi Ovadya in the Molecular Cell Biology Department, were recently published in Nature Communications.
The research began with an investigation into the way that the immune system is involved in a crucial activity: clearing away old, senescent cells that spell trouble for the body when they hang around. Senescent cells – not completely dead but suffering loss of function or irreparable damage – have been implicated in diseases of aging by promoting inflammation. The researchers used mice in which a crucial gene for this immune activity was missing. At two years (elderly, for mice), the bodies of these mice had a greater accumulation of senescent cells compared with the mice in which the gene for removing these cells was intact. The mice missing the gene suffered from chronic inflammation, and various functions in their bodies appeared to be diminished. They also looked older – and died earlier – than their normal counterparts.
Next, the researchers gave the mice a drug that inhibits the function of certain proteins that help the aging cells survive in their senescent state, to see if this would contribute to the removal of these cells from the body. The drugs were administered to mice whose aging was a result of the malfunctions the group had uncovered in the immune system as well as those suffering premature aging from a different genetic error. The treated mice responded exceptionally well to the drug: Their blood tests and activity tests showed improvement, and their tissues appeared to be much closer to those of young mice. The scientists counted senescent cells, finding many fewer of them remaining in the treated mice’s bodies; and when they looked for signs of inflammation, they found that this, too, was significantly lower. The mice treated with the drug were more active and their median lifespan rose.
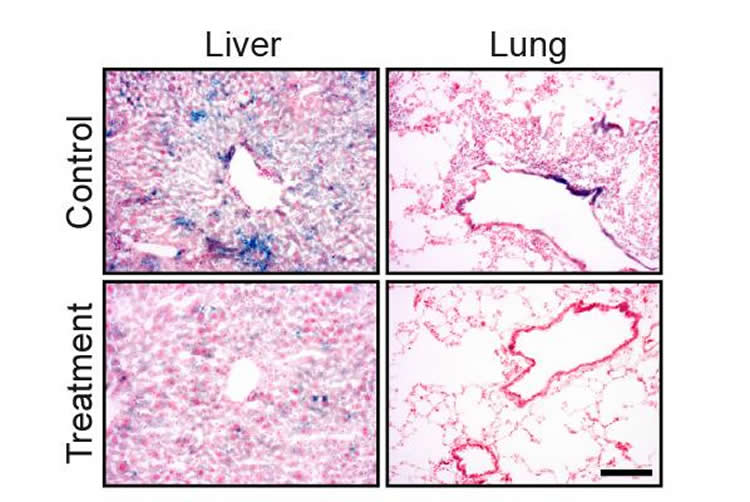
The scientists intend to continue exploring ways to prompt the human body to remove its old senescent cells, particularly to find means of activating the immune system to do this job. That is, if future experimentation proves their theories correct, they could end up creating truly “anti-aging” therapies.
Funding: Prof. Valery Krizhanovsky’s research is supported by the Sagol Institute for Longevity Research; the Ilse Katz Institute for Material Sciences and Magnetic Resonance Research; the Rising Tide Foundation; the Quinquin Foundation; Mr. and Mrs. Bruce Kanter; and the European Research Council.
Source: Yael Edelman – Weizmann Institute of Science
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Weizmann Institute of Science.
Original Research: Open access research for “Impaired immune surveillance accelerates accumulation of senescent cells and aging” by Yossi Ovadya, Tomer Landsberger, Hanna Leins, Ezra Vadai, Hilah Gal, Anat Biran, Reut Yosef, Adi Sagiv, Amit Agrawal, Alon Shapira, Joseph Windheim, Michael Tsoory, Reinhold Schirmbeck, Ido Amit, Hartmut Geiger & Valery Krizhanovsky in Nature Communications. Published December 21 2018.
doi:10.1038/s41467-018-07825-3
[cbtabs][cbtab title=”MLA”]Weizmann Institute of Science”The Immune System’s Fountain of Youth.” NeuroscienceNews. NeuroscienceNews, 31 December 2018.
<https://neurosciencenews.com/immune-system-youth-10402/>.[/cbtab][cbtab title=”APA”]Weizmann Institute of Science(2018, December 31). The Immune System’s Fountain of Youth. NeuroscienceNews. Retrieved December 31, 2018 from https://neurosciencenews.com/immune-system-youth-10402/[/cbtab][cbtab title=”Chicago”]Weizmann Institute of Science”The Immune System’s Fountain of Youth.” https://neurosciencenews.com/immune-system-youth-10402/ (accessed December 31, 2018).[/cbtab][/cbtabs]
Abstract
Impaired immune surveillance accelerates accumulation of senescent cells and aging
Cellular senescence is a stress response that imposes stable cell-cycle arrest in damaged cells, preventing their propagation in tissues. However, senescent cells accumulate in tissues in advanced age, where they might promote tissue degeneration and malignant transformation. The extent of immune-system involvement in regulating age-related accumulation of senescent cells, and its consequences, are unknown. Here we show that Prf1−/− mice with impaired cell cytotoxicity exhibit both higher senescent-cell tissue burden and chronic inflammation. They suffer from multiple age-related disorders and lower survival. Strikingly, pharmacological elimination of senescent-cells by ABT-737 partially alleviates accelerated aging phenotype in these mice. In LMNA+/G609G progeroid mice, impaired cell cytotoxicity further promotes senescent-cell accumulation and shortens lifespan. ABT-737 administration during the second half of life of these progeroid mice abrogates senescence signature and increases median survival. Our findings shed new light on mechanisms governing senescent-cell presence in aging, and could motivate new strategies for regenerative medicine.







