Summary: Using brain organoid models, researchers have identified how the brain grows much larger and has three times as many neurons, as the brains of chimpanzees and gorillas.
Source: UK Research and Innovation
A new study is the first to identify how human brains grow much larger, with three times as many neurons, compared with chimpanzee and gorilla brains. The study, led by researchers at the Medical Research Council (MRC) Laboratory of Molecular Biology in Cambridge, UK, identified a key molecular switch that can make ape brain organoids grow more like human organoids, and vice versa.
The study, published in the journal Cell, compared ‘brain organoids’ – 3D tissues grown from stem cells which model early brain development – that were grown from human, gorilla and chimpanzee stem cells.
Similar to actual brains, the human brain organoids grew a lot larger than the organoids from other apes.
Dr Madeline Lancaster, from the MRC Laboratory of Molecular Biology, who led the study, said: “This provides some of the first insight into what is different about the developing human brain that sets us apart from our closest living relatives, the other great apes. The most striking difference between us and other apes is just how incredibly big our brains are.”
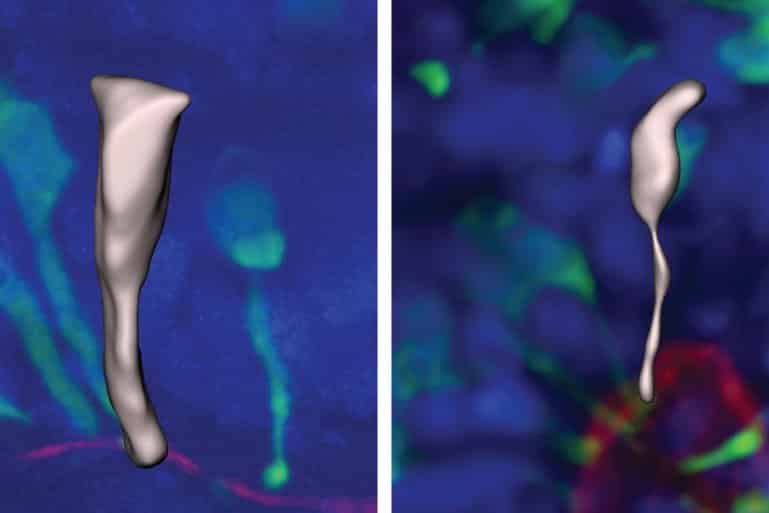
During the early stages of brain development, neurons are made by stem cells called neural progenitors. These progenitor cells initially have a cylindrical shape that makes it easy for them to split into identical daughter cells with the same shape.
The more times the neural progenitor cells multiply at this stage, the more neurons there will be later.
As the cells mature and slow their multiplication, they elongate, forming a shape like a stretched ice-cream cone.
Previously, research in mice had shown that their neural progenitor cells mature into a conical shape and slow their multiplication within hours.
Now, brain organoids have allowed researchers to uncover how this development happens in humans, gorillas and chimpanzees.
They found that in gorillas and chimpanzees this transition takes a long time, occurring over approximately five days.
Human progenitors were even more delayed in this transition, taking around seven days. The human progenitor cells maintained their cylinder-like shape for longer than other apes and during this time they split more frequently, producing more cells.
This difference in the speed of transition from neural progenitors to neurons means that the human cells have more time to multiply. This could be largely responsible for the approximately three-fold greater number of neurons in human brains compared with gorilla or chimpanzee brains.
Dr Lancaster said: “We have found that a delayed change in the shape of cells in the early brain is enough to change the course of development, helping determine the numbers of neurons that are made.
“It’s remarkable that a relatively simple evolutionary change in cell shape could have major consequences in brain evolution. I feel like we’ve really learnt something fundamental about the questions I’ve been interested in for as long as I can remember – what makes us human.”
To uncover the genetic mechanism driving these differences, the researchers compared gene expression – which genes are turned on and off – in the human brain organoids versus the other apes.
They identified differences in a gene called ‘ZEB2’, which was turned on sooner in gorilla brain organoids than in the human organoids.
To test the effects of the gene in gorilla progenitor cells, they delayed the effects of ZEB2. This slowed the maturation of the progenitor cells, making the gorilla brain organoids develop more similarly to human – slower and larger.
Conversely, turning on the ZEB2 gene sooner in human progenitor cells promoted premature transition in human organoids, so that they developed more like ape organoids.
The researchers note that organoids are a model and, like all models, do not to fully replicate real brains, especially mature brain function. But for fundamental questions about our evolution, these brain tissues in a dish provide an unprecedented view into key stages of brain development that would be impossible to study otherwise.
Dr Lancaster was part of the team that created the first brain organoids in 2013.
Funding: This study was funded by the Medical Research Council, European Research Council and Cancer Research UK.
About this evolutionary neuroscience research news
Source: UK Research and Innovation
Contact: Hilary Jones – UK Research and Innovation
Image: The image is credited to S.Benito-Kwiecinski/MRC LMB/Cell
Original Research: Open access.
“An early cell shape transition drives evolutionary expansion of the human forebrain” by Madeline Lancaster et al. Cell
Abstract
An early cell shape transition drives evolutionary expansion of the human forebrain
Highlights
- •Human brain organoids are expanded relative to nonhuman apes prior to neurogenesis
- •Ape neural progenitors go through a newly identified transition morphotype state
- •Delayed morphological transition with shorter cell cycles underlie human expansion
- •ZEB2 is as an evolutionary regulator of this transition
Summary
The human brain has undergone rapid expansion since humans diverged from other great apes, but the mechanism of this human-specific enlargement is still unknown.
Here, we use cerebral organoids derived from human, gorilla, and chimpanzee cells to study developmental mechanisms driving evolutionary brain expansion.
We find that neuroepithelial differentiation is a protracted process in apes, involving a previously unrecognized transition state characterized by a change in cell shape. Furthermore, we show that human organoids are larger due to a delay in this transition, associated with differences in interkinetic nuclear migration and cell cycle length. Comparative RNA sequencing (RNA-seq) reveals differences in expression dynamics of cell morphogenesis factors, including ZEB2, a known epithelial-mesenchymal transition regulator.
We show that ZEB2 promotes neuroepithelial transition, and its manipulation and downstream signaling leads to acquisition of nonhuman ape architecture in the human context and vice versa, establishing an important role for neuroepithelial cell shape in human brain expansion.






