Summary: The retrieval of mitochondria via a feedback loop is vital to sustaining synaptic transmission.
Source: NIH
Our thoughts, feelings, and movements are controlled by billions of neurons talking to each other at trillions of specialized communication points called synapses. In an in-depth study of neurons grown in laboratory petri dishes, National Institutes of Health researchers discovered how the chattiest of some synapses find the energy to support intense conversations thought to underlie learning and memory.
Their results, published in Nature Metabolism, suggest that a series of chemical reactions control a feedback loop that senses the need for more energy and replenishes it by recruiting cellular powerplants, called mitochondria, to the synapses.
The experiments were performed by researchers in a lab led by Zu-Hang Sheng, Ph.D., at the NIH’s National Institute of Neurological Disorders and Stroke (NINDS).
The team studied synapses that use the neurotransmitter glutamate to communicate. Communication happens when a packet of glutamate is released from presynaptic boutons which are tiny protrusions that stick out, like beads on a string, of long, wiry parts of neurons called axons.
Previously, Dr. Sheng’s team showed that synaptic communication is an energy-demanding process and that mitochondria traveling along axons can control signals sent by boutons. Boutons that had mitochondria sent stronger and more consistent signals than those that were missing powerplants. The difference was due to higher energy levels produced by the mitochondria in the form of ATP.
In this study, led by Sunan Li, Ph.D., a post-doctoral fellow at NINDS, the team investigated what happens when boutons undergo intense communication thought to underlie learning and memory.
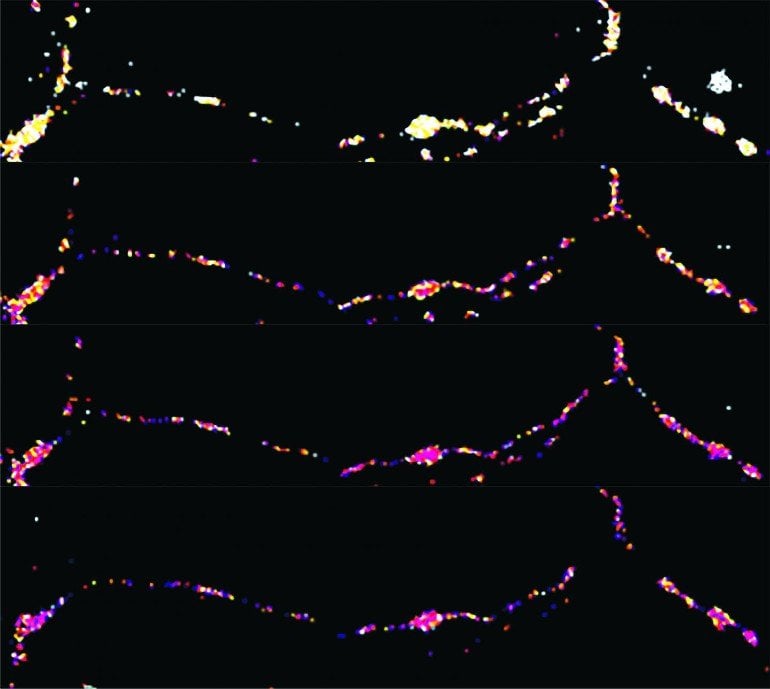
They found that this type of signaling quickly dropped energy levels at boutons. These changes triggered a series of chemical reactions controlled by an energy sensor called AMP-activated protein kinases (AMPK) that ultimately led to the rapid recruitment of mitochondria to the boutons. Genetically blocking or chemically interfering with this feedback loop prevented the delivery of mitochondria to boutons and lowered energy levels. This, in turn, reduced synaptic responses during intense communication more than seen in control cells and slowed the recovery of the responses after the bursts ended.
The researchers concluded that this feedback loop may normally play a critical role in providing the energy needed to sustain synaptic communication throughout a healthy nervous system. For example, they cite studies which implied that problems with this system may occur in some cases of Alzheimer’s disease and other neurological disorders.
Funding: This study was supported by the NIH’s NINDS Division of Intramural Research (NS003029, NS002946).
About this neuroscience research news
Source: NIH
Contact: Christopher G. Thomas – NIH
Image: The image is credited to Sheng lab NINDS/NIH.
Original Research: Closed access.
“The cross-talk of energy sensing and mitochondrial anchoring sustains synaptic efficacy by maintaining presynaptic metabolism” by Li, S. et al. Nature Metabolism.
Abstract
The cross-talk of energy sensing and mitochondrial anchoring sustains synaptic efficacy by maintaining presynaptic metabolism
Mitochondria supply ATP essential for synaptic transmission. Neurons face exceptional challenges in maintaining energy homoeostasis at synapses. Regulation of mitochondrial trafficking and anchoring is critical for neurons to meet increased energy consumption during sustained synaptic activity. However, mechanisms recruiting and retaining presynaptic mitochondria in sensing synaptic ATP levels remain elusive. Here we reveal an energy signalling axis that controls presynaptic mitochondrial maintenance. Activity-induced presynaptic energy deficits can be rescued by recruiting mitochondria through the AMP-activated protein kinase (AMPK)–p21-activated kinase (PAK) energy signalling pathway. Synaptic activity induces AMPK activation within axonal compartments and AMPK–PAK signalling triggers phosphorylation of myosin VI, which drives mitochondrial recruitment and syntaphilin-mediated anchoring on presynaptic filamentous actin. This pathway maintains presynaptic energy supply and calcium clearance during intensive synaptic activity. Disrupting this signalling cross-talk triggers local energy deficits and intracellular calcium build-up, leading to impaired synaptic efficacy during trains of stimulation and reduced recovery from synaptic depression after prolonged synaptic activity. Our study reveals a mechanistic cross-talk between energy sensing and mitochondria anchoring to maintain presynaptic metabolism, thus fine-tuning short-term synaptic plasticity and prolonged synaptic efficacy.






