Summary: Researchers watch, in real time, as immune cells help repair the meninges following mild traumatic brain injury.
Source: NIH/NINDS.
Following head injury, the protective lining that surrounds the brain may get a little help from its friends: immune cells that spring into action to assist with repairs. In a new study, scientists from the National Institutes of Health watched in real-time as different immune cells took on carefully timed jobs to fix the damaged lining of the brain, also known as meninges, in mice. These results may help provide clues to the discovery that the meninges in humans may heal following mild traumatic brain injury (mTBI) and why additional hits to the head can be so devastating.
“The lining of the brain, with help from the immune system, has a remarkable ability to put itself back together again after injury,” said Dorian McGavern, Ph.D., scientist at the NIH’s National Institute of Neurological Disorders and Stroke and the senior author of the study published in Nature Immunology. “As we learn more about all the cells involved in the repair process, we may be able to identify potential targets for therapy that lead to better outcomes for patients.”
The study came about from an observation on MRI scans of adult patients who experienced a concussion or mTBI. Around half of patients with mTBI show evidence of injury to blood vessels in the meninges, which appears on MRI scans as a vascular dye leaking out of the damaged vessels.
The meninges are a collection of membranes that line the central nervous system and help protect brain and spinal cord tissue from various forms of injury. Damage to the meninges can cause cell death in underlying brain tissue.
Dr. McGavern’s team found that while most patients had repaired their leaky blood vessels within 20 days, 17 percent of patients still showed leakage on their MRI scans three months after injury, indicating ongoing meningeal damage and incomplete recovery.
To learn more about the recovery process, Dr. McGavern and his group used state-of-the-art imaging tools to watch, in real-time, what happened in the mouse meninges up to one week after injury. They also developed a method of analyzing where immune cells gathered in the damaged meninges during the repair process.
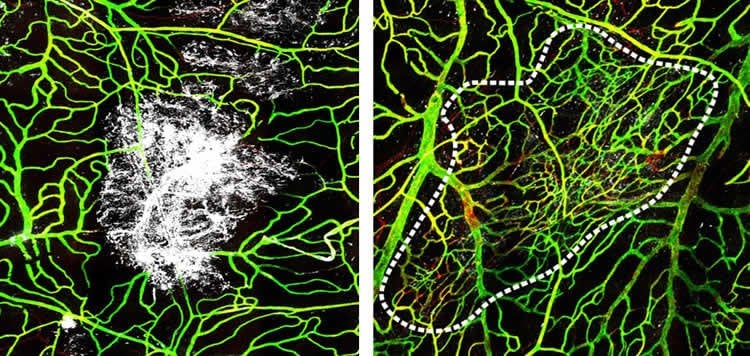
Within the first day of injury, immune cells from the blood called inflammatory monocytes entered the core of the injured meningeal tissue and started clearing away dead cells. These cells were assisted a few days later by a different type of blood monocyte that worked around the lesion edge to help rebuild damaged blood vessels, which were completely restored and fully functional within a week. Dr. McGavern and his team found that the actions of these different immune cell types did not overlap and that blocking the activity of one did not cause the other to take over.
“Following a head injury, the meninges call in a clean-up crew, followed by a separate repair crew, to help fix damaged blood vessels,” said Dr. McGavern.
The researchers also found that the timing of a second head injury has a significant impact on the repair process in mice. A second injury experienced within one day of the first TBI led to additional inflammation and the wound healing phase of repair, during which blood vessels are fixed, did not occur. However, if the re-injury occurred after a few days, once the wound healing phase had already begun, there was no effect on the meningeal repair process and blood vessels were rebuilt normally.
“The timing of a second head injury may determine whether the meninges can be repaired. We have shown on a cellular level, that two or more head injuries within a very short amount of time can have really dire consequences for the brain lining and its ability to repair,” said Dr. McGavern. “It is possible that patients who did not fully recover following a head injury may have had problems with the first phase of the repair process.”
Experiments revealed that the molecule matrix metalloproteinase 2 (Mmp2) may play a critical role in the restoration of blood vessels. The wound-healing immune cells release Mmp2, which breaks down the matrix, or glue, holding cells together, allowing room for blood vessels to be rebuilt. When Mmp2 was blocked, there was a large decrease in the number of vessels that were repaired.
Further research is needed to uncover additional molecules and genes involved in the repair processes and identify ways to speed up the course of recovery following head injury.
Funding: This work was supported by the NIH Intramural Research Program.
Source: NIH/NINDS
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Dorian McGavern, Ph.D., National Institute of Neurological Disorders and Stroke.
Video Source: Video credited to NIH/NINDS.
Original Research: Abstract for “Distinct myeloid cell subsets promote meningeal remodeling and vascular repair after mild traumatic brain injury” by Matthew V. Russo, Lawrence L. Latour & Dorian B. McGavern in Nature Immunology. Published April 16 2018.
doi:10.1038/s41590-018-0086-2
[cbtabs][cbtab title=”MLA”]NIH/NINDS “Researchers Watch Brain’s Lining Heal After Head Injury.” NeuroscienceNews. NeuroscienceNews, 16 April 2018.
<https://neurosciencenews.com/head-injury-brain-lining-8809/>.[/cbtab][cbtab title=”APA”]NIH/NINDS (2018, April 16). Researchers Watch Brain’s Lining Heal After Head Injury. NeuroscienceNews. Retrieved April 16, 2018 from https://neurosciencenews.com/head-injury-brain-lining-8809/[/cbtab][cbtab title=”Chicago”]NIH/NINDS “Researchers Watch Brain’s Lining Heal After Head Injury.” https://neurosciencenews.com/head-injury-brain-lining-8809/ (accessed April 16, 2018).[/cbtab][/cbtabs]
Abstract
Distinct myeloid cell subsets promote meningeal remodeling and vascular repair after mild traumatic brain injury
Mild traumatic brain injury (mTBI) can cause meningeal vascular injury and cell death that spreads into the brain parenchyma and triggers local inflammation and recruitment of peripheral immune cells. The factors that dictate meningeal recovery after mTBI are unknown at present. Here we demonstrated that most patients who had experienced mTBI resolved meningeal vascular damage within 2–3 weeks, although injury persisted for months in a subset of patients. To understand the recovery process, we studied a mouse model of mTBI and found extensive meningeal remodeling that was temporally reliant on infiltrating myeloid cells with divergent functions. Inflammatory myelomonocytic cells scavenged dead cells in the lesion core, whereas wound-healing macrophages proliferated along the lesion perimeter and promoted angiogenesis through the clearance of fibrin and production of the matrix metalloproteinase MMP-2. Notably, a secondary injury experienced during the acute inflammatory phase aborted this repair program and enhanced inflammation, but a secondary injury experienced during the wound-healing phase did not. Our findings demonstrate that meningeal vasculature can undergo regeneration after mTBI that is dependent on distinct myeloid cell subsets.