Summary: Researchers have identified how the ASPM gene controls the growth of the cerebral cortex. They report mutations in this gene can reduce brain volume by half in human microcephaly cases.
Source: Yale.
Scientists have identified several genes that when mutated can cause children to be born with microcephaly, or abnormally small heads. But exactly how these genes control brain size has remained unclear. A team of researchers headed by scientists at Yale and Harvard have identified how one such gene controls the growth of the cerebral cortex, the seat of higher-order thinking in humans.
The gene, abnormal spindle-like microcephaly-associated (ASPM), helps keep neural progenitor cells proliferating for a longer period in the developing embryo, which in turn leads to production of more neurons and supporting glial cells, the researchers report April 11 in the journal Nature.
Mutations in ASPM reduces brain volume by half in human microcephaly cases.
Previous efforts to study the function of genes involved in brain growth have been hampered by differences in species. For instance, brains of mice are 1/1000th the size of humans and lack the complex folds that dramatically expand the surface area of the brain. In the new study, the researchers tracked changes in the brains of ferrets, which share many structural and molecular similarities with higher-order mammals such as primates. Ferrets lacking the ASPM gene show strikingly similar brain abnormalities found in human microcephaly patients, such as 25%-40% reduction in brain volume, with the most severe reduction in the frontal cortex.
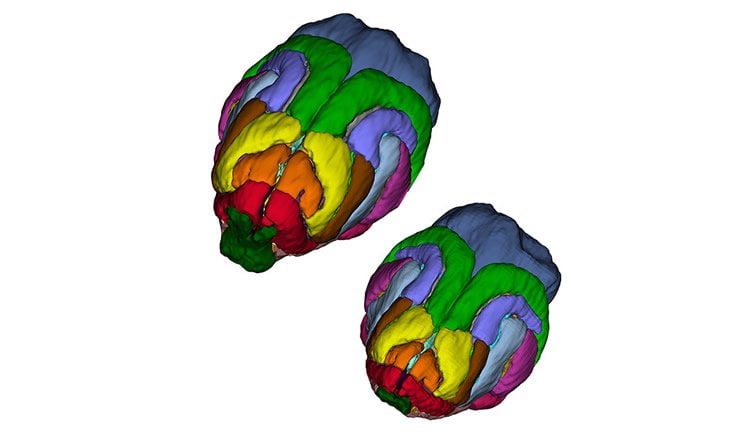
Also the mutant ferret cortex shows a massive premature displacement of a specific neural progenitor cell that has been implicated in expansion of the cerebral cortex in primates.
“ASPM seems to make neural progenitor cells linger in the germinal zone, allowing them to divide more in place,” said Yale’s Byoung-Il Bae, associate research scientist in neurosurgery and co-corresponding author of the paper.
The evolution of the ASPM protein is significantly associated with brain size in primates, whales and dolphins. “Perhaps the same developmental mechanism led to expanded brain size in both primates and cetaceans,” Bae said.
Harvard’s Christopher A. Walsh of Boston’s Children Hospital is co-corresponding author of the paper.
Funding: The research was funded by National Institutes of Health, Howard Hughes Medical Institute, and Allen Discovery Center program.
Source: Bill Hathaway – Yale
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Yale Magnetic Resonance Research Center.
Original Research: Abstract for “Aspm knockout ferret reveals an evolutionary mechanism governing cerebral cortical size” by Matthew B. Johnson, Xingshen Sun, Andrew Kodani, Rebeca Borges-Monroy, Kelly M. Girskis, Steven C. Ryu, Peter P. Wang, Komal Patel, Dilenny M. Gonzalez, Yu Mi Woo, Ziying Yan, Bo Liang, Richard S. Smith, Manavi Chatterjee, Daniel Coman, Xenophon Papademetris, Lawrence H. Staib, Fahmeed Hyder, Joseph B. Mandeville, P. Ellen Grant, Kiho Im, Hojoong Kwak, John F. Engelhardt, Christopher A. Walsh & Byoung-Il Bae in Nature. Published April 9 2018.
doi:10.1038/s41586-018-0035-0
[cbtabs][cbtab title=”MLA”]Yale “One Gene Spurred Growth of Key Brain Area.” NeuroscienceNews. NeuroscienceNews, 15 April 2018.
<https://neurosciencenews.com/genetics-cerebral-cortex-8797/>.[/cbtab][cbtab title=”APA”]Yale (2018, April 15). One Gene Spurred Growth of Key Brain Area. NeuroscienceNews. Retrieved April 15, 2018 from https://neurosciencenews.com/genetics-cerebral-cortex-8797/[/cbtab][cbtab title=”Chicago”]Yale “One Gene Spurred Growth of Key Brain Area.” https://neurosciencenews.com/genetics-cerebral-cortex-8797/ (accessed April 15, 2018).[/cbtab][/cbtabs]
Abstract
Aspm knockout ferret reveals an evolutionary mechanism governing cerebral cortical size
The human cerebral cortex is distinguished by its large size and abundant gyrification, or folding. However, the evolutionary mechanisms that drive cortical size and structure are unknown. Although genes that are essential for cortical developmental expansion have been identified from the genetics of human primary microcephaly (a disorder associated with reduced brain size and intellectual disability), studies of these genes in mice, which have a smooth cortex that is one thousand times smaller than the cortex of humans, have provided limited insight. Mutations in abnormal spindle-like microcephaly-associated (ASPM), the most common recessive microcephaly gene, reduce cortical volume by at least 50% in humans but have little effect on the brains of mice this probably reflects evolutionarily divergent functions of ASPM. Here we used genome editing to create a germline knockout of Aspm in the ferret (Mustela putorius furo), a species with a larger, gyrified cortex and greater neural progenitor cell diversity than mice, and closer protein sequence homology to the human ASPM protein. Aspm knockout ferrets exhibit severe microcephaly (25–40% decreases in brain weight), reflecting reduced cortical surface area without significant change in cortical thickness, as has been found in human patients, suggesting that loss of ‘cortical units’ has occurred. The cortex of fetal Aspm knockout ferrets displays a very large premature displacement of ventricular radial glial cells to the outer subventricular zone, where many resemble outer radial glia, a subtype of neural progenitor cells that are essentially absent in mice and have been implicated in cerebral cortical expansion in primates. These data suggest an evolutionary mechanism by which ASPM regulates cortical expansion by controlling the affinity of ventricular radial glial cells for the ventricular surface, thus modulating the ratio of ventricular radial glial cells, the most undifferentiated cell type, to outer radial glia, a more differentiated progenitor.