Summary: Researchers at UBC believe they have identified a method that could help stop Alzheimer’s disease in its tracks.
Source: UBC.
How one amino acid can spell difference between a healthy aging brain and early onset Alzheimer’s disease.
The old real estate adage about “location, location, location” might also apply to the biochemical genesis of Alzheimer’s disease, according to new research from the University of British Columbia.
Scientists had previously identified a couple of crucial steps in the formation of a protein called amyloid beta, which accumulates in clumps, or “plaques,” in the brains of people with Alzheimer’s disease. Those discoveries inspired efforts at disrupting the biochemical carving of amyloid beta’s precursor protein into its final, toxic shape.
The latest drugs being tested try to silence an enzyme, called BACE1, that cuts the precursor protein. But BACE1 has other functions that are beneficial, so stopping it altogether could bring unwanted side effects — including disrupting the production of myelin, the protective insulation of brain cells.
Psychiatry Professor and Alzheimer’s researcher Weihong Song has found that changing where the cut is made — in effect, guiding the enzyme’s scissors to a different point — could achieve the same goal, with less collateral damage.
Song built upon two discoveries in the past decade of two rare mutations: one found in Italian people that leads to early onset Alzheimer’s disease, and another found in Icelandic people that staves off Alzheimer’s disease.
Dr. Song’s team at UBC’s Townsend Family Laboratories was particularly intrigued by the diametrically opposite effects of both mutations because they affected the same point on the precursor protein’s chain of 770 amino acids, swapping one acid for another.
As Dr. Song and his team describe in the July 19 issue of the Journal of Neuroscience, they injected one set of mice with a virus carrying the Italian gene mutation, and another set with the Icelandic mutation.
They found that the amino acid substitution affected where the precursor protein was cleaved. The Icelandic mutation resulted in a shortened form of amyloid beta, which does not become “sticky” and turn into plaque. The Italian mutation produced a longer, “stickier” version of amyloid beta, which ultimately becomes neuron-smothering plaque.
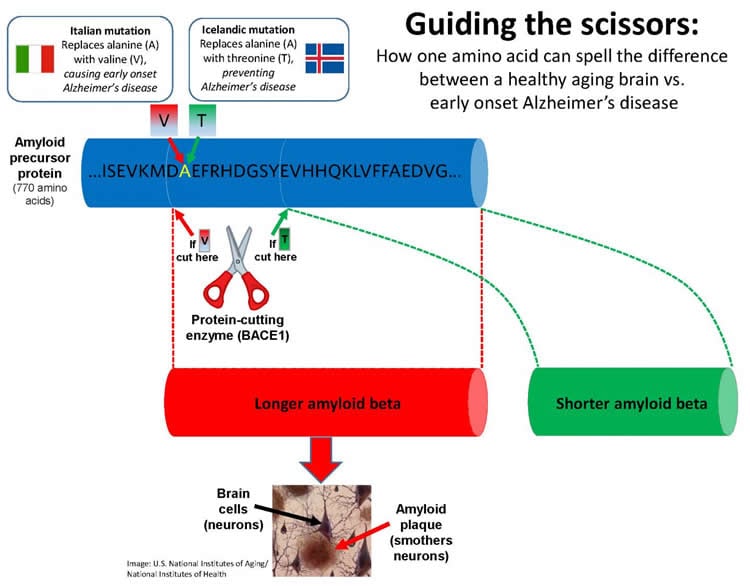
Actually, the effects were a matter of degree: Each mutation led to more cuts in one location or more cuts in the other location. But in the gradual degradation of Alzheimer’s disease, that could be enough – reducing the levels of the offending toxin could translate into many more years of life before cognitive decline sets in.
“If we can adjust where BACE1 cuts the precursor protein, we will have a very precise way of slowing the build-up of plaque in the brain, without affecting other processes,” says Dr. Song, a Canada Research Chair in Alzheimer’s Disease and the Jack Brown and Family Professor. “This provides a new target in our search for a drug – instead of sledgehammer, it’s more like a scalpel.”
Funding: This research was supported by Canadian Institutes of Health Research, Alzheimer’s Society of Canada, University of British Columbia.
Source: Brian Kladko – UBC
Image Source: NeuroscienceNews.com image is credited to Brian Kladko and Weihong Song/University of British Columbia.
Original Research: Abstract for “BACE1 Cleavage Site Selection Critical for Amyloidogenesis and Alzheimer’s Pathogenesis” by Shuting Zhang, Zhe Wang, Fang Cai, Mingming Zhang, Yili Wu, Jing Zhang and Weihong Song in Journal of Neuroscience. Published online July 19 2017 doi:10.1523/JNEUROSCI.0340-17.2017
[cbtabs][cbtab title=”MLA”]UBC “Steering an Enzyme’s ‘Scissors’ Shows Potential for Stopping Alzheimer’s .” NeuroscienceNews. NeuroscienceNews, 19 July 2017.
<https://neurosciencenews.com/enzyme-stop-alzheimers-7129 />.[/cbtab][cbtab title=”APA”]UBC (2017, July 19). Steering an Enzyme’s ‘Scissors’ Shows Potential for Stopping Alzheimer’s . NeuroscienceNew. Retrieved July 19, 2017 from https://neurosciencenews.com/enzyme-stop-alzheimers-7129 /[/cbtab][cbtab title=”Chicago”]UBC “Steering an Enzyme’s ‘Scissors’ Shows Potential for Stopping Alzheimer’s .” https://neurosciencenews.com/enzyme-stop-alzheimers-7129 / (accessed July 19, 2017).[/cbtab][/cbtabs]
Abstract
BACE1 Cleavage Site Selection Critical for Amyloidogenesis and Alzheimer’s Pathogenesis
Mutations in amyloid β precursor protein (APP) gene alter APP processing, either causing familial Alzheimer’s disease (AD) or protecting against dementia. Under normal conditions, β-site APP cleaving enzyme 1 (BACE1) cleaves APP at minor Asp1 site to generate C99 for amyloid β protein (Aβ) production, and predominantly at major Glu11 site to generate C89, resulting in truncated Aβ production. We discovered that A673V mutation, the only recessive AD-associated APP mutation, shifted the preferential β-cleavage site of BACE1 in APP from the Glu11 site to the Asp1 site both in male and female transgenic mice in vivo and in cell lines and primary neuronal culture derived from timed pregnant rats in vitro, resulting in a much higher C99 level and C99/C89 ratio. All other mutations at this site, including the protective Icelandic A673T mutation, reduced C99 generation, and decreased the C99/C89 ratio. Furthermore, A673V mutation caused stronger dimerization between mutant and wild-type APP, enhanced the lysosomal degradation of the mutant APP, and inhibited γ-secretase cleavage of the mutant C99 to generate Aβ, leading to recessively inherited AD. The results demonstrate that APP673 regulates APP processing and the BACE1 cleavage site selection is critical for amyloidogenesis in AD pathogenesis, and implicate a pharmaceutical potential for targeting the APP673 site for AD drug development.
SIGNIFICANCE STATEMENT β-site APP cleaving enzyme 1 (BACE1) is essential for amyloid β protein production. We discovered that A673V mutation shifted the BACE1 cleavage site from the Glu11 to the Asp1 site, resulting in much higher C99 level and C99/C89 ratio. All other mutations at this site of amyloid β precursor protein (APP) reduced C99 generation and decreased the C99/C89 ratio. Furthermore, A673V mutation resulted in stronger dimerization between mutant and wild-type APP, enhanced the lysosomal degradation of the mutant APP, and inhibited γ-secretase cleavage of the mutant C99 to generate amyloid β protein, leading to recessively inherited Alzheimer’s disease (AD). The results demonstrate that APP673 regulates APP processing, and the BACE1 cleavage site selection is critical for amyloidogenesis in AD pathogenesis, and implicate a pharmaceutical potential for targeting the APP673 site for AD drug development.
“BACE1 Cleavage Site Selection Critical for Amyloidogenesis and Alzheimer’s Pathogenesis” by Shuting Zhang, Zhe Wang, Fang Cai, Mingming Zhang, Yili Wu, Jing Zhang and Weihong Song in Journal of Neuroscience. Published online July 19 2017 doi:10.1523/JNEUROSCI.0340-17.2017






