Summary: Researchers have identified a potential new drug target for COVID-19. The scientists mapped the anatomical structures of two critical SARS-CoV-2 proteins in the nsp10/nsp16 complex. If a drug can be developed to inhibit nsp10/nsp16, researchers believe the immune system should be able to detect the infection and eradicate it faster.
Source: UCR
A new potential drug target has been identified in SARS CoV-2 — the virus that causes COVID-19 — by scientists at the Center of Structural Genomics of Infectious Diseases, or CSGID, who say multiple drugs will be needed to treat the pandemic.
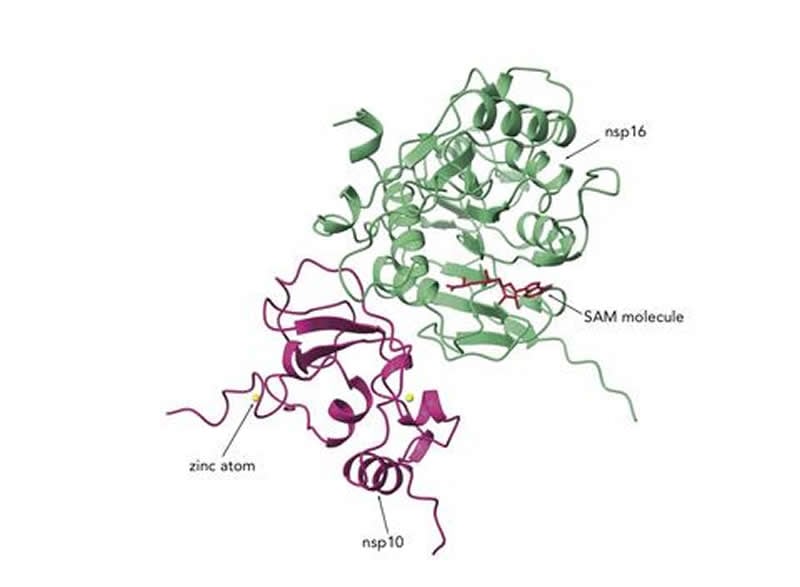
CSGID scientists mapped the atomic structures of two critical SARS-CoV-2 proteins in a complex called nsp10/16. CSGID is an international consortium of scientists investigating the structure of the coronavirus proteins to in aid drug development, and includes Adam Godzik, a professor of biomedical sciences at University of California, Riverside’s School of Medicine.
The two proteins modify the genetic material of the virus to make it look more like the human RNA, allowing it to avoid host anti-viral defenses and giving it time to multiply. The researchers believe if a drug can be developed to inhibit nsp10/nsp16, the immune system should be able to detect the virus and eradicate it faster.
“We first designed the constructs — pieces of DNA— to express the two proteins,” Godzik said. “Subsequently, we analyzed the structure of these proteins.”
Protein production, purification, crystallization and structure determination work for the drug test was performed at Northwestern University, the University of Chicago, and Argonne National Laboratory.
Godzik said the research team is releasing the protein structures to the public, so other research groups can use them for drug discovery efforts.
“This is a really beautiful target, because it’s a protein absolutely essential for the virus to replicate,” said lead investigator Karla Satchell, a professor of microbiology-immunology at Northwestern and CSGID director.
Satchell’s team is sending the new protein complex to Purdue University, the drug-discovery site of the center, to be screened for novel inhibitors that could be developed as new drugs.
The nsp10/nsp16 protein complex is called an RNA methyltransferase or MTase. It is comprised of two proteins bound together. The association of the two pieces together is required to make a functional protein, according to prior research on SARS.
This is the fourth protein structure of potential drug target of SARS-CoV-2 determined by the CSGID team of scientists.
“We need multiple drugs to treat this virus, because this disease is likely to be with us for a long time,” Satchell said. “It’s not good enough for us to develop a single drug. If COVID-19 develops a resistance to one drug, then we need others.”
Structures of three other proteins important for the replication of the virus were also released: the nsp15 endonuclease, nsp3 ADP ribose phosphate, and nsp9 replicase. These structures were determined by the CSGID scientists working at the University of Chicago, headed by Professor Andrzej Joachimiak, a distinguished fellow at Argonne, and an adjunct professor at Northwestern. All work conducted by both the University of Chicago and Northwestern teams was designed by Godzik’s bioinformatic team, based on research conducted on SARS.
“This is all part of an effort to map the entire protein structural repertoire on the new virus,” Godzik said. “Expanding the structural coverage by solving additional structures is the most immediate follow-up direction. The second direction is co-crystallization experiments with potential drugs; we want to know if and how the coronavirus proteins bind to them. This would help to improve the drugs, making them better geared toward this particular pathogen.”
CSGID is racing to release more structures for drug development. The center’s goal is to determine structures of all of the proteins that are potential drug targets. The team is also collaborating to provide proteins to investigators for designing improved vaccines.
“The center has shown a great ability to bring structure biology to the scientific community at an unprecedented rate,” Satchell said.
The center’s work has become more challenging because many universities have reduced activities and some labs have shut down entirely.
“Our ability to do experiments is abating,” Satchell said. “Still, the center will continue to release new structures until we reach our goal.”
CSGID is funded as a contract from the National Institute of Allergy and Infectious Diseases, or NIAID, part of the National Institutes of Health, in part to serve as a response site to conduct structure biology research in the event of an unexpected infectious disease outbreak. The NIAID has been working closely with CSGID since early January to coordinate center activities with other research supported by NIAID and to share information from the center with NIAID personnel specializing in drug discovery.
Funding: This study has been funded by contract HHSN272201700060C from the National Institute of Allergy and Infectious Diseases.
Source:
UCR
Media Contacts:
UCR News – UCR
Image Source:
The image is credited to Northwestern University.
Original Research: Open access
“Crystal structure of Nsp15 endoribonuclease NendoU from SARS-CoV-2”. Youngchang Kim, Robert Jedrzejczak, Natalia I. Maltseva, Michael Endres, Adam Godzik, Karolina Michalska, Andrzej Joachimiak.
bioRxiv doi:10.1101/2020.03.02.968388.
Abstract
Crystal structure of Nsp15 endoribonuclease NendoU from SARS-CoV-2
Severe Acute Respiratory Syndrome Coronavirus 2 is rapidly spreading around the world. There is no existing vaccine or proven drug to prevent infections and stop virus proliferation. Although this virus is similar to human and animal SARS- and MERS-CoVs the detailed information about SARS-CoV-2 proteins structures and functions is urgently needed to rapidly develop effective vaccines, antibodies and antivirals. We applied high-throughput protein production and structure determination pipeline at the Center for Structural Genomics of Infectious Diseases to produce SARS-CoV-2 proteins and structures. Here we report the high-resolution crystal structure of endoribonuclease Nsp15/NendoU from SARS-CoV-2 – a virus causing current world-wide epidemics. We compare this structure with previously reported models of Nsp15 from SARS and MERS coronaviruses.






