Summary: Researchers report that without enough copper, fat builds up in fat cells without being used.
Source: UC Berkeley.
Copper puts a brake on a brake of fat burning, suggesting a possible role in obesity, diabetes.
Is copper deficiency contributing to the obesity epidemic?
Though small amounts of copper are essential to health – oysters, liver, beans and nuts are good sources – copper’s role in metabolism has been unclear: Some studies found that it boosted fat burning, others that it depressed it.
University of California, Berkeley, Lawrence Berkeley National Laboratory and Howard Hughes Medical Institute researchers have now clarified the critical role that copper plays in nutrition: It helps move fat out of fat cells – called adipocytes – and into the blood stream for use as energy.
Without enough copper, fat builds up in fat cells without being utilized, said Christopher Chang, the Class of 1942 Chair and a professor of chemistry and of molecular and cell biology at UC Berkeley.
“Unlike other studies that link copper levels both to increased or decreased fat metabolism, our study shows definitively how it works – it’s a signal that turns on fat cells,” said Chang, who also is a faculty scientist at Berkeley Lab and a Howard Hughes Medical Institute investigator. “If we could find a way to burn fat more efficiently, this could be a big contribution to dealing with obesity and diabetes.”
The new study appeared online this week, and will be published in the July print issue of the journal Nature Chemical Biology.
The findings point not only to a new role for copper in metabolism, but highlight the key role copper plays throughout the body. Previously, Chang showed that copper is a key signaling molecule in the brain, tamping down over-excitation of nerve cells.
“The work we have done with copper really shows that signaling is not restricted to just a few elements on the periodic table, like sodium, calcium and potassium,” he said. “Copper is a brand new class; in a sense we have basically added a fourth letter to the alphabet, in terms of metal elements that contribute to the language of signaling.”
The similar roles copper plays in regulating nerve cells and fat burning highlight a growing perception that the neurological system, specifically the brain, plays a role in diseases of metabolism and the immune system, such as obesity and inflammation.
“Some of us are now thinking about obesity as a neurological disease rather than strictly a metabolic disease, because there are potentially connections between your fat tissue and the brain,” he said. “The notion that obesity and obesity-related diseases like diabetes may be neurological in nature opens the door to new basic science and therapeutic approaches.”
Chang cautions against ingesting copper supplements as a result of these studies, however. Too much copper can lead to imbalances in other essential minerals, including zinc.
Copper-related diseases
With a longstanding interest in the biological roles of metals, Chang focused on fat metabolism after reading that copper is often given to cows to regulate weight gain. To discover how copper actually works in fat metabolism, he chose to study mice with a genetic defect that produces symptoms similar to those of a rare human illness, Wilson’s disease. In both situations, a key enzyme that moves copper in and out of cells is mutated, causing misregulation and copper overload – often to toxic levels – in the liver. The condition is also characterized by bloated fat cells.
He and his team, in particular postdocs Lakshmi Krishnamoorthy and Joseph Cotruvo Jr., discovered that in the mice, copper is hoarded by the liver, basically starving fat cells of copper so that they cannot properly regulate fat storage and burning in organs such as the stomach and pancreas. This overload is accompanied by lower than normal levels of fats, or lipids, in the blood.
“Lipolysis is the breakdown of large pieces of fat into smaller pieces so they can circulate in the blood and be burned throughout the body,” he said. “We discovered that in these Wilson’s mice, low copper in the fat cells made them unable to burn fat as well as normal mice.”
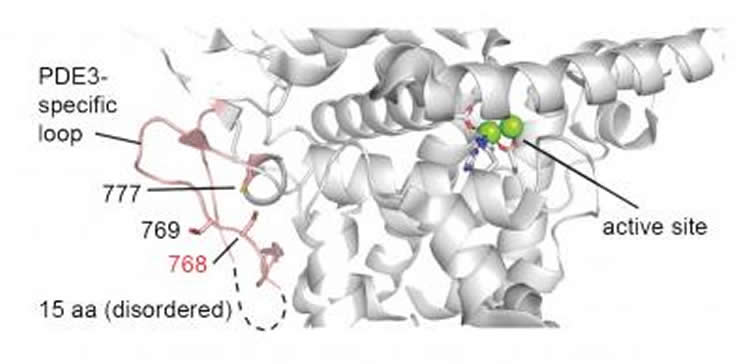
They succeeded in tracking down exactly how copper works: It releases a brake on fat burning. Normally, a second messenger molecule called cyclic-AMP (cAMP) activates the enzymes that break down the fat molecules. They found, however, that another enzyme (phosphodiesterase 3, or PDE3) blocks cAMP, probably to prevent fat breakdown when it is not needed, such as when we’re couch potatoes. Copper blocks this enzyme, thereby “putting a brake on a brake,” Chang said.
Chang is continuing his study of copper’s roles in the brain and in fat metabolism, looking for possible ways to target the copper pathway to treat neurodegenerative brain diseases or diseases of metabolism. He also is looking at copper’s roles in perception and sleep.
These studies have led him also to explore the connections between the central nervous system – the brain and spinal cord – and the peripheral nervous system, which innervates all our organs, and how that may cause disease.
Funding: The work was funded by the National Institutes of Health (GM 79465, GM067166, GM101502)..
Source: Robert Sanders – UC Berkeley
Image Source: This NeuroscienceNews.com image is credited to Lakshmipriya Krishnamoorthy and Joseph Cotruvo Jr., UC Berkeley.
Original Research: Abstract for “Copper regulates cyclic-AMP-dependent lipolysis” by Lakshmi Krishnamoorthy, Joseph A Cotruvo Jr, Jefferson Chan, Harini Kaluarachchi, Abigael Muchenditsi, Venkata S Pendyala, Shang Jia, Allegra T Aron, Cheri M Ackerman, Mark N Vander Wal, Timothy Guan, Lukas P Smaga, Samouil L Farhi, Elizabeth J New, Svetlana Lutsenko and Christopher J Chang in Nature Chemical Biology. Published online June 6 2016 doi:10.1038/nchembio.2098
[cbtabs][cbtab title=”MLA”]UC Berkeley. “Copper is Essential for Burning Fat.” NeuroscienceNews. NeuroscienceNews, 10 June 2016.
<https://neurosciencenews.com/copper-obesity-fat-diabetes-4434/>.[/cbtab][cbtab title=”APA”]UC Berkeley. (2016, June 10). Copper is Essential for Burning Fat. NeuroscienceNews. Retrieved June 10, 2016 from https://neurosciencenews.com/copper-obesity-fat-diabetes-4434/[/cbtab][cbtab title=”Chicago”]UC Berkeley. “Copper is Essential for Burning Fat.” https://neurosciencenews.com/copper-obesity-fat-diabetes-4434/ (accessed June 10, 2016).[/cbtab][/cbtabs]
Abstract
Copper regulates cyclic-AMP-dependent lipolysis
Cell signaling relies extensively on dynamic pools of redox-inactive metal ions such as sodium, potassium, calcium and zinc, but their redox-active transition metal counterparts such as copper and iron have been studied primarily as static enzyme cofactors. Here we report that copper is an endogenous regulator of lipolysis, the breakdown of fat, which is an essential process in maintaining body weight and energy stores. Using a mouse model of genetic copper misregulation, in combination with pharmacological alterations in copper status and imaging studies in a 3T3-L1 white adipocyte model, we found that copper regulates lipolysis at the level of the second messenger, cyclic AMP (cAMP), by altering the activity of the cAMP-degrading phosphodiesterase PDE3B. Biochemical studies of the copper-PDE3B interaction establish copper-dependent inhibition of enzyme activity and identify a key conserved cysteine residue in a PDE3-specific loop that is essential for the observed copper-dependent lipolytic phenotype.
“Copper regulates cyclic-AMP-dependent lipolysis” by Lakshmi Krishnamoorthy, Joseph A Cotruvo Jr, Jefferson Chan, Harini Kaluarachchi, Abigael Muchenditsi, Venkata S Pendyala, Shang Jia, Allegra T Aron, Cheri M Ackerman, Mark N Vander Wal, Timothy Guan, Lukas P Smaga, Samouil L Farhi, Elizabeth J New, Svetlana Lutsenko and Christopher J Chang in Nature Chemical Biology. Published online June 6 2016 doi:10.1038/nchembio.2098






