Summary: A newly developed model that mimics the blood-brain barrier could help researchers more efficiently test drugs to treat a range of neurological diseases.
Source: University of Portsmouth.
Scientists have helped provide a way to better understand how to enable drugs to enter the brain and how cancer cells make it past the blood brain barrier.
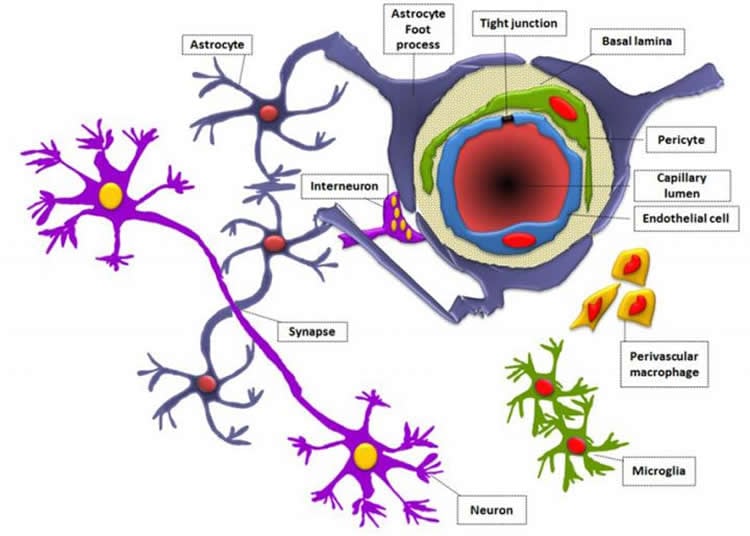
The brain is protected by the near-impermeable blood brain barrier, a fortress which protects the brain but which also prevents the treatment of brain diseases, including brain tumours.
Dr Zaynah Maherally and team at the University of Portsmouth have developed a model that mimics the blood brain barrier, which could pave the way for better, more efficient and reliable tests of drugs to treat brain diseases.
The model, the result of slow painstaking research started in 2007, is published in the FASEB Journal.
Dr Maherally said: “The blood brain barrier is strikingly complex and notoriously difficult for scientists to breach. Its role, to protect the brain, makes it difficult for most drugs to make their way into the brain to treat brain tumours.
“It is a dynamic structural, physiological and biochemical fortification that, in essence, protects the brain by providing multiple layers of armour, stopping molecules from entering the brain. It’s highly selective in what it allows past.
“In a person with a brain disease like a brain tumour, or other neurological conditions such as Parkinson’s disease, dementia or head injuries, the strikingly complex defence works against medics and scientists trying to deliver what could be life-saving or life-prolonging treatments.”
A common way medics currently gain access to the brain is by using a pump to deliver drugs into the brain using a catheter, bypassing the blood brain barrier.
The researchers’ major goal was to develop a 3D all-human reproducible and reliable model of the blood brain barrier using human cells in order to better simulate the human blood brain barrier for the study of diseases and treatments.
Many researchers use non-human animal cells to build blood brain barrier models with which to explore brain cancer and deliver drugs to the brain.
Animal models have been relied upon until now as there wasn’t a reliable alternative and accessing human tissue for research can be difficult. Professor Geoff Pilkington, who leads the research group at Portsmouth, is delighted at the progress that’s been made in modelling the blood brain barrier, a holy grail in the study of brain diseases.
He said: “This is the first real, 3D, all-human blood brain barrier model and it’s hugely significant in our field.”
Research will now widen, he said, to better understand how cancers metastasize from breast and lung to the brain as well as evaluating nano-particle drug delivery and making opportunities to create temporary openings in the barrier to allow drugs to pass through into the brain.
Dr Maherally said: “It’s taken several years to get to this stage and we believe this model will significantly reduce the number of animals used in such studies and reduce the time it takes to get a promising therapeutic into clinical trials.”
Funding: The research was funded by Animal Free Research UK, previously known as Dr Hadwen Trust, and is supported by the charity Brain Tumour Research.
Source: Kate Daniell – University of Portsmouth
Image Source: NeuroscienceNews.com image is credited to Samah Jassam.
Original Research: Abstract for “Real-time acquisition of transendothelial electrical resistance in an all-human, in vitro, 3-dimensional, blood–brain barrier model exemplifies tight-junction integrity” by Zaynah Maherally, Helen L. Fillmore, Sim Ling Tan, Suk Fei Tan, Samah A. Jassam, Friederike I. Quack, Kathryn E. Hatherell, and Geoffrey J. Pilkington in FASEB Journal. Published online September 7 2017 doi:10.1096/fj.201700162R
[cbtabs][cbtab title=”MLA”]University of Portsmouth “New Model May Help Researchers Overcome the Brain’s Fortress-Like Barrier.” NeuroscienceNews. NeuroscienceNews, 19 September 2017.
<https://neurosciencenews.com/bbb-model-7524/>.[/cbtab][cbtab title=”APA”]University of Portsmouth (2017, September 19). New Model May Help Researchers Overcome the Brain’s Fortress-Like Barrier. NeuroscienceNews. Retrieved September 19, 2017 from https://neurosciencenews.com/bbb-model-7524/[/cbtab][cbtab title=”Chicago”]University of Portsmouth “New Model May Help Researchers Overcome the Brain’s Fortress-Like Barrier.” https://neurosciencenews.com/bbb-model-7524/ (accessed September 19, 2017).[/cbtab][/cbtabs]
Abstract
Real-time acquisition of transendothelial electrical resistance in an all-human, in vitro, 3-dimensional, blood–brain barrier model exemplifies tight-junction integrity
The blood–brain barrier (BBB) consists of endothelial cells, astrocytes, and pericytes embedded in basal lamina (BL). Most in vitro models use nonhuman, monolayer cultures for therapeutic-delivery studies, relying on transendothelial electrical resistance (TEER) measurements without other tight-junction (TJ) formation parameters. We aimed to develop reliable, reproducible, in vitro 3-dimensional (3D) models incorporating relevant human, in vivo cell types and BL proteins. The 3D BBB models were constructed with human brain endothelial cells, human astrocytes, and human brain pericytes in mono-, co-, and tricultures. TEER was measured in 3D models using a volt/ohmmeter and cellZscope. Influence of BL proteins—laminin, fibronectin, collagen type IV, agrin, and perlecan—on adhesion and TEER was assessed using an electric cell-substrate impedance–sensing system. TJ protein expression was assessed by Western blotting (WB) and immunocytochemistry (ICC). Perlecan (10 µg/ml) evoked unreportedly high, in vitro TEER values (1200 Ω) and the strongest adhesion. Coculturing endothelial cells with astrocytes yielded the greatest resistance over time. ICC and WB results correlated with resistance levels, with evidence of prominent occludin expression in cocultures. BL proteins exerted differential effects on TEER, whereas astrocytes in contact yielded higher TEER values and TJ expression.—Maherally, Z., Fillmore, H. L., Tan, S. L., Tan, S. F., Jassam, S. A., Quack, F. I., Hatherell, K. E., Pilkington, G. J. Real-time acquisition of transendothelial electrical resistance in an all-human, in vitro, 3-dimensional, blood–brain barrier model exemplifies tight-junction integrity.
“Real-time acquisition of transendothelial electrical resistance in an all-human, in vitro, 3-dimensional, blood–brain barrier model exemplifies tight-junction integrity” by Zaynah Maherally, Helen L. Fillmore, Sim Ling Tan, Suk Fei Tan, Samah A. Jassam, Friederike I. Quack, Kathryn E. Hatherell, and Geoffrey J. Pilkington in FASEB Journal. Published online September 7 2017 doi:10.1096/fj.201700162R






