Summary: Mutations to the EGR1 gene disrupt social behaviors in zebrafish models of ASD. Additionally, the mutation disrupts dopamine signaling from specific neurons, contributing to mood and social behavior disorders.
Source: University of Oregon
Zebrafish are social creatures. When they see another member of their species, they’ll orient towards them and swim closer, much like a human at a cocktail party turning to face someone who’s telling a joke over a plate of hors d’oeuvres.
A mutation in a gene called EGR1 snuffs out this social behavior in zebrafish, researchers in the UO’s Institute of Neuroscience show in a new study. And it disrupts dopamine signaling from certain neurons in the brain, which can affect mood and social behavior.
In humans, mutations in the EGR1 gene have been linked to mental health conditions like schizophrenia and depression, and they also appear to play a role in autism. So understanding more about how this gene shapes social behavior could help researchers unravel the biological basis for a number of complex conditions that have strong social components.
The team, led by UO biology professors Philip Washbourne and Judith Eisen, reports their findings in a paper published April 6 in the journal eNeuro.
To test the effects of EGR1, the researchers put pairs of zebrafish in adjacent tanks. Fish could see each other but couldn’t sense any water movement or chemical signals from their neighboring fish.
Fish with normally functioning EGR1 tended to swim closer to the barrier and orient towards the other fish when it approached, the researchers found. But fish with a mutation in both copies of the EGR1 gene didn’t show particular interest in their piscine neighbor. They didn’t get as close, and they didn’t position themselves towards the glass in the same way.
The researchers also measured the fish’s response to moving dots that shift and dart like shadows of another fish. As they had in the first experiments, zebrafish with normal EGR1 responded socially to those dots as if they were another fish. Fish with the mutated gene didn’t.
“This is giving a small piece of the brain circuits involved in social behavior,” Washbourne said. “From there, we can go upstream and downstream and try to put the circuit together.”
For instance, the EGR1 gene controls a gene that makes tyrosine hydroxylase, a chemical that the body needs to make dopamine. Fish with the EGR1 mutation make less of this chemical in certain neurons in one part of their brain.
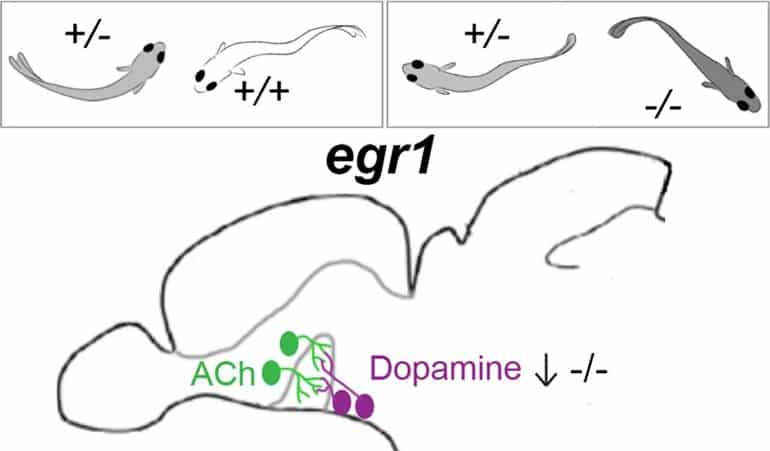
Without as much tyrosine hydroxylase, there’s less dopamine, too. So a domino effect started by one gene can send ripple effects through circuits in the brain that affect mood and social behavior.
Social behavior is far more complex than one gene or brain circuit, but Eisen and Washbourne see value in looking at the most basic social behaviors in that way. The neurons they studied are in a brain region that exists not just in humans but also in many other animals with much simpler brains.
Human brains layer more complexity on top of a basic ground plan that’s shared across the brains of many species, Eisen said.
“You can look at these brain regions in other animals where the cortex is not as complex as in humans and learn a great deal,” she said.
For example, here, researchers linked EGR1 to one specific social behavior in zebrafish: following another fish. In mice or humans, that same gene, and the related brain circuits, might direct a different social behavior.
“Different species can use the same cells to take on different social tasks,” Washbourne said.
About this genetics and ASD research news
Author: Lauren Hamers
Source: University of Oregon
Contact: Lauren Hamers – University of Oregon
Image: The image is credited to the researchers
Original Research: Open access.
“Egr1 Is Necessary for Forebrain Dopaminergic Signaling during Social Behavior” by Alexandra Tallafuss et al. eNeuro
Abstract
Egr1 Is Necessary for Forebrain Dopaminergic Signaling during Social Behavior
Finding the link between behaviors and their regulatory molecular pathways is a major obstacle in treating neuropsychiatric disorders. The immediate early gene (IEG) EGR1 is implicated in the etiology of neuropsychiatric disorders, and is linked to gene pathways associated with social behavior.
Despite extensive knowledge of EGR1 gene regulation at the molecular level, it remains unclear how EGR1 deficits might affect the social component of these disorders.
Here, we examined the social behavior of zebrafish with a mutation in the homologous gene egr1.
Mutant fish exhibited reduced social approach and orienting, whereas other sensorimotor behaviors were unaffected. On a molecular level, expression of the dopaminergic biosynthetic enzyme, tyrosine hydroxylase (TH), was strongly decreased in TH-positive neurons of the anterior parvocellular preoptic nucleus. These neurons are connected with basal forebrain (BF) neurons associated with social behavior.
Chemogenetic ablation of around 30% of TH-positive neurons in this preoptic region reduced social attraction to a similar extent as the egr1 mutation.
These results demonstrate the requirement of Egr1 and dopamine signaling during social interactions, and identify novel circuitry underlying this behavior.






