Summary: According to a new study, alcohol increases neural activity in the central amygdala.
Source: Scripps Research Institute.
A new study led by scientists at The Scripps Research Institute (TSRI) could help researchers develop personalized treatments for alcoholism and alcohol use disorder.
The research reveals a key difference between the brains of alcohol-dependent versus nondependent rats. When given alcohol, both groups showed increased activity in a region of the brain called the central amygdala (CeA) — but this activity was due to two completely different brain signaling pathways.
TSRI Professor Marisa Roberto, senior author of the new study, said the findings could help researchers develop more personalized treatments for alcohol dependence, as they evaluate how a person’s brain responds to different therapeutics.
The findings were published recently online ahead of print in The Journal of Neuroscience.
Researchers Find Brain’s Alcohol Response ‘Switch’
The new research builds on the Roberto lab’s previous discovery that alcohol increases neuronal activity in the CeA. The researchers found increased activity both nondependent, or naïve, and alcohol-dependent rats.
As they investigated this phenomenon in the new study, Roberto and her colleagues were surprised to find that the mechanisms underlying this increased activity differed between the two groups.
By giving naïve rats a dose of alcohol, the researchers engaged proteins called calcium channels and increased neuronal activity. Neurons fired as the specific calcium channels at play, called L-type voltage-gated calcium channels (LTCCs), boosted the release of a neurotransmitter called GABA. Blocking these LTCCs reduced voluntary alcohol consumption in naïve rats.
But in alcohol-dependent rats, the researchers found decreased abundance of LTCCs on neuronal cell membranes, disrupting their normal ability to drive a dose of alcohol’s effects on CeA activity. Instead, increased neuronal activity was driven by a stress hormone called corticotropin-releasing factor (CRF) and its type 1 receptor (CRF1). The researchers found that blocking CeA CRF1s reduced voluntary alcohol consumption in the dependent rats.
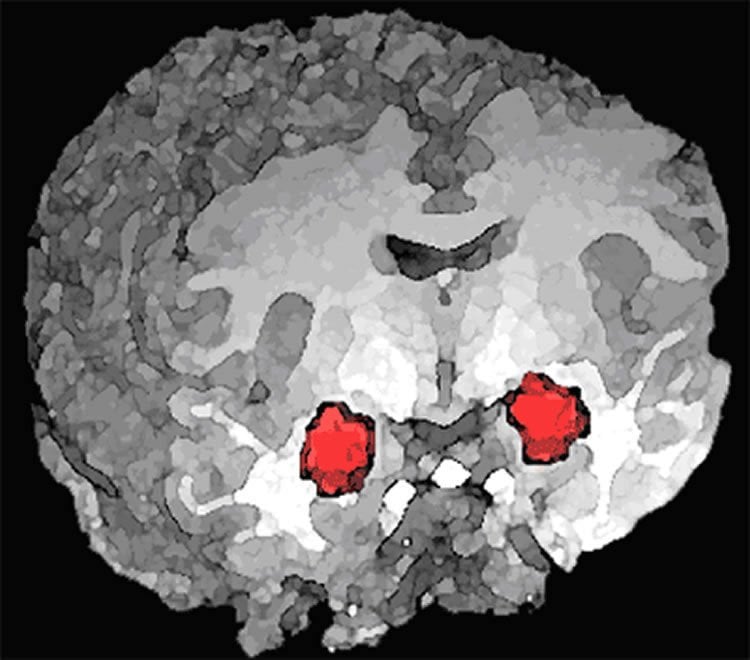
Studying these two groups shed light on how alcohol functionally alters the brain, Roberto explained.
“There is a switch in the molecular mechanisms underlying the CeA’s response to alcohol (from LTCC- to CRF1-driven) as the individual transitions to the alcohol-dependent state,” she said.
The cellular and molecular experiments were led by TSRI Research Associate and study first author Florence Varodayan. The behavioral tests were conducted by TSRI Research Associate Giordano de Guglielmo in the lab of TSRI Associate Professor Olivier George.
Roberto hopes the findings lead to better ways to treat alcohol dependence. Alcohol use disorder appears to have many different root causes, but the new findings suggest doctors could analyze certain symptoms or genetic markers to determine which patients are likely to have CRF-CRF1 hyperactivation and benefit from the development of a novel drug that blocks that activity.
In addition to Roberto, Varodayan and de Guglielmo, authors of the study, “Alcohol dependence disrupts amygdalar L-type voltage-gated calcium channel mechanisms,” were Marian Logrip and Olivier George of TSRI.
Funding: The study was supported by the National Institutes of Health (grants AA015566, AA021491, AA017447, AA006420, AA013498, AA020608, AA022977 and AA021802).
Source: Madeline McCurry-Schmidt – Scripps Research Institute
Image Source: NeuroscienceNews.com image is in the public domain.
Original Research: Abstract for “Alcohol dependence disrupts amygdalar L-type voltage-gated calcium channel mechanisms” by Florence P. Varodayan, Giordano de Guglielmo, Marian L. Logrip, Olivier George and Marisa Roberto in Journal of Neuroscience. Published online March 31 2017 doi:10.1523/JNEUROSCI.3721-16.2017
[cbtabs][cbtab title=”MLA”]Scripps Research Institute “Surprising Brain Change Appears to Drive Alcohol Dependence.” NeuroscienceNews. NeuroscienceNews, 12 April 2017.
<https://neurosciencenews.com/alcohol-dependence-amygdala-6399/>.[/cbtab][cbtab title=”APA”]Scripps Research Institute (2017, April 12). Surprising Brain Change Appears to Drive Alcohol Dependence. NeuroscienceNew. Retrieved April 12, 2017 from https://neurosciencenews.com/alcohol-dependence-amygdala-6399/[/cbtab][cbtab title=”Chicago”]Scripps Research Institute “Surprising Brain Change Appears to Drive Alcohol Dependence.” https://neurosciencenews.com/alcohol-dependence-amygdala-6399/ (accessed April 12, 2017).[/cbtab][/cbtabs]
Abstract
Alcohol dependence disrupts amygdalar L-type voltage-gated calcium channel mechanisms
L-type voltage-gated calcium channels (LTCCs) are implicated in several psychiatric disorders that are co-morbid with alcoholism and involve amygdala dysfunction. Within the amygdala, the central nucleus (CeA) is critical in acute alcohol’s reinforcing actions, and its dysregulation in human alcoholics drives their negative emotional state and motivation to drink. Here we investigated the specific role of CeA LTCCs in the effects of acute alcohol at the molecular, cellular physiology and behavioral levels, and their potential neuroadaptation in alcohol-dependent rats. Alcohol increases CeA activity (neuronal firing rates and GABA release) in naïve rats by engaging LTCCs, and intra-CeA LTCC blockade reduces alcohol intake in non-dependent rats. Alcohol dependence reduces CeA LTCC membrane abundance and disrupts this LTCC-based mechanism; instead, corticotropin-releasing factor type 1 receptors (CRF1s) mediate alcohol’s effects on CeA activity and drive the escalated alcohol intake of alcohol-dependent rats. Collectively, our data indicate that alcohol dependence functionally alters the molecular mechanisms underlying the CeA’s response to alcohol (from LTCC- to CRF1-driven). This mechanistic switch contributes to and reflects the prominent role of the CeA in the negative emotional state that drives excessive drinking.
SIGNIFICANCE STATEMENT
The central amygdala (CeA) plays a critical role in the development of alcohol dependence. As a result, much preclinical alcohol research aims to identify relevant CeA neuroadaptions that promote the transition to dependence. Here we report that acute alcohol increases CeA neuronal activity in naïve rats by engaging L-type calcium channels (LTCCs) and that intra-CeA LTCC blockade reduces alcohol intake in non-dependent rats. Alcohol dependence disrupts this LTCC-based mechanism; instead, corticotropin-releasing factor type 1 receptors (CRF1s) mediate alcohol’s effects on CeA activity and drive the escalated alcohol intake of alcohol-dependent rats. This switch reflects the important role of the CeA in the pathophysiology of alcohol dependence and represents a new potential avenue for therapeutic intervention during the transition period.
“Alcohol dependence disrupts amygdalar L-type voltage-gated calcium channel mechanisms” by Florence P. Varodayan, Giordano de Guglielmo, Marian L. Logrip, Olivier George and Marisa Roberto in Journal of Neuroscience. Published online March 31 2017 doi:10.1523/JNEUROSCI.3721-16.2017






