Summary: Researchers shed light on how behavior is organized in the brain. The study reports learned behaviors are organized in a hierarchy of multiple levels of control.
Source: Salk Institute.
Driving to work, typing an email or playing a round of golf—people perform actions such as these throughout the day. But neuroscientists are still unsure how the brain orchestrates complex actions or switches to a new action—behaviors that are impaired in disorders such as Parkinson’s disease or obsessive-compulsive disorder (OCD).
Now, Salk researchers have resolved a longstanding scientific debate about how behavior is organized in the brain. Led by Associate Professor Xin Jin, the team discovered that learned behavior is organized in a hierarchy with multiple levels of control, offering possible new therapeutic targets for disorders that involve an inability to control one’s actions. The work, which appeared in the journal Cell on June 28, 2018, utilized mice trained to perform complex action sequences to make the discovery.
“For many decades, scientists have been debating how the brain organizes behavior,” says Jin, the study’s senior author. “Using optogenetics, a technique that uses light to manipulate brain cells’ activity, we were able to change individual actions that the animals were planning to perform, revealing this precise level of neural control.”
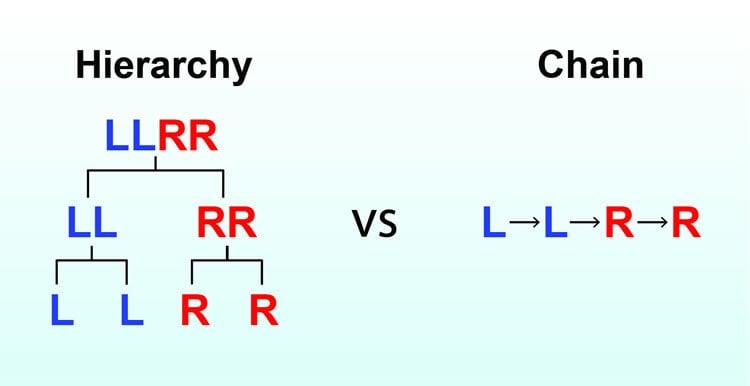
When you learn a new behavior—such as tying your shoelaces—a brain region called the striatum orchestrates the series of actions: bending down, grabbing the laces, and tying the knot. Scientists have long debated whether these types of steps are organized in a chain, with each step triggering the next (like falling dominoes) or whether there is a more hierarchical system at work, with multiple levels of control (like an office organization chart).
Over several weeks, Jin’s team trained mice to carry out a series of lever presses. In a custom-built box with a lever to the left and a lever to the right, the mice learned that pressing the levers in the specific order of left-left-right-right yielded a treat. (The team named this series “the penguin dance” after a line dance sometimes performed at weddings or parties.)
While the mice performed the penguin dance sequence, a computer recorded the activity of two specific types of brain cells: D1 and D2 neurons, which make up the majority of cells in the striatum and have been implicated in learning and performing actions. The researchers used optogenetics to activate these neurons with laser light and diphtheria toxin to inactivate them, allowing the team to identify and isolate how D1 and D2 cells control behavior.
Interestingly, stimulating D1 neurons caused the mice to add one additional lever press to the sequence, while stimulating D2 neurons caused the mice to skip the next lever presses.
By manipulating D1 or D2 neurons at precise points during performance of the penguin dance, the researchers were able to piece together how the sequence was learned and organized by the brain. They also discovered unexpected mixtures of the neurons working in tandem to drive or suppress behavior.
“Neurons are like snowflakes,” says UC San Diego graduate student and Salk researcher Claire Geddes, the paper’s first author. “D1 and D2 neurons have certain similar patterns, but they don’t all do exactly the same thing. There’s a complexity to how they work together to control movement.”
The team found evidence for three levels of control in neuronal activity. The lowest level represented activity with each individual step in the action sequence, while the highest level represented activity only when starting or stopping the overall sequence. At an intermediate level, neurons were active only during the animals’ switch from one action to another. It’s akin to an employee’s supervisor overseeing every task versus a senior executive who mostly monitors whether a project was begun or completed. In between are the organization’s middle managers, operating amid the other two.
According to Jin, Geddes and coauthor Hao Li, the work reveals a subtle complexity to neuronal behavior in the striatum that may help explain why the learned actions we perform remain so flexible—our brain’s supervisors, middle managers and senior executives can update our behaviors to respond to challenges in our ever-changing environment.
Adds Jin, “I’m particularly excited about this study because it solves a long-standing debate for a fundamental question in behavioral neuroscience. At the same time, by identifying how different cell types in the brain control our behavior, it provides new insight into potential treatments for different neurological diseases.”
Funding: The work was funded by the US National Institutes of Health (R01NS083815 and R01AG047669), the Dana Foundation, Ellison Medical Foundation and Whitehall Foundation.
Source: Salk Institute
Publisher: Organized by NeuroscienceNews.com.
Image Source: NeuroscienceNews.com image is credited to Salk Institute.
Video Source: Video credited to Salk Institute.
Original Research: Abstract for “Optogenetic Editing Reveals the Hierarchical Organization of Learned Action Sequences” by Claire E. Geddes, Hao Li, and Xin Jin in Cell. Published June 28 2018.
doi:10.1016/j.cell.2018.06.012
[cbtabs][cbtab title=”MLA”]Salk Institute “Insights Into Amyloids Associated with Alzheimer’s and Type 2 Diabetes.” NeuroscienceNews. NeuroscienceNews, 28 June 2018.
<https://neurosciencenews.com/action-orgnization-9472/>.[/cbtab][cbtab title=”APA”]Salk Institute (2018, June 28). Insights Into Amyloids Associated with Alzheimer’s and Type 2 Diabetes. NeuroscienceNews. Retrieved June 28, 2018 from https://neurosciencenews.com/action-orgnization-9472/[/cbtab][cbtab title=”Chicago”]Salk Institute “Insights Into Amyloids Associated with Alzheimer’s and Type 2 Diabetes.” https://neurosciencenews.com/action-orgnization-9472/ (accessed June 28, 2018).[/cbtab][/cbtabs]
Abstract
Alzheimer’s disease pathology propagation by exosomes containing toxic amyloid-beta oligomers
Highlights
•Non-back-propagation learning of sequences depends on striatal NMDA receptors
•Striatal direct pathway facilitates actions and controls sequence start/stop
•Striatal indirect pathway inhibits actions and mediates subsequence switch
•Optogenetic manipulations unveil the hierarchical structure of learned sequences
Summary
The organization of action into sequences underlies complex behaviors that are essential for organismal survival and reproduction. Despite extensive studies of innate sequences in relation to central pattern generators, how learned action sequences are controlled and whether they are organized as a chain or a hierarchy remain largely unknown. By training mice to perform heterogeneous action sequences, we demonstrate that striatal direct and indirect pathways preferentially encode different behavioral levels of sequence structure. State-dependent closed-loop optogenetic stimulation of the striatal direct pathway can selectively insert a single action element into the sequence without disrupting the overall sequence length. Optogenetic manipulation of the striatal indirect pathway completely removes the ongoing subsequence while leaving the following subsequence to be executed with the appropriate timing and length. These results suggest that learned action sequences are not organized in a serial but rather a hierarchical structure that is distinctly controlled by basal ganglia pathways.






